Optimizing HTE Batch Reactor Design for High-Throughput Organic Synthesis: A Guide for Research Scientists
This article provides a comprehensive guide to High-Throughput Experimentation (HTE) batch reactor design for accelerating organic synthesis and drug discovery.
Optimizing HTE Batch Reactor Design for High-Throughput Organic Synthesis: A Guide for Research Scientists
Abstract
This article provides a comprehensive guide to High-Throughput Experimentation (HTE) batch reactor design for accelerating organic synthesis and drug discovery. Targeted at researchers and development professionals, it explores the foundational principles driving HTE adoption, details practical reactor configurations and experimental methodologies, addresses common troubleshooting and optimization challenges, and validates performance through comparisons with traditional methods. The scope covers integrating automation, data analysis, and predictive modeling to enhance efficiency and success rates in compound library synthesis, reaction optimization, and route scouting.
Why HTE Batch Reactors Are Revolutionizing Organic Synthesis
Within the paradigm of High-Throughput Experimentation (HTE) batch reactor design for organic synthesis research, the central impediment to realizing the full potential of accelerated discovery is no longer conceptual imagination but experimental velocity and the generation of high-fidelity, interpretable data. This whitepaper examines the core technical challenges and solutions at the intersection of automation, data acquisition, and informatics that define the modern synthetic laboratory.
The Data Bottleneck in HTE Workflows
The throughput of parallelized micro-batch reactors has surpassed our traditional capacity for reaction setup, analysis, and data management.
Table 1: Comparative Throughput and Data Output of Synthesis Platforms
| Platform Type | Typical Reactor Count | Experiments/Week (Theoretical) | Data Points/Experiment | Primary Bottleneck |
|---|---|---|---|---|
| Traditional Round-Bottom Flask | 1-10 | 5-20 | ~3 (TLC, Yield, Purity) | Manual Operations |
| Automated Serial Flow | 1 (continuous) | 50-200 | 10-50 (Online Analytics) | Parameter Exploration |
| HTE Batch Array (Modern) | 24 - 384 | 500 - 5000 | 100+ (HPLC/MS, NMR) | Sample Processing & Data Structuring |
Key Experimental Protocols for Accelerated HTE
Protocol 2.1: Automated Reaction Assembly & Execution
Objective: To prepare and initiate a 96-well plate of catalytic cross-coupling reactions with varied ligands and bases.
- Plate Design: A CSV file defining the experiment is loaded into the robotic control software. The design includes variations in ligand (8 choices), base (6 choices), and concentration (2 levels) with randomized well assignments to mitigate positional effects.
- Solid Dispensing: An automated solid dispenser portions precisely weighed catalyst (Pd-source, 0.5-2.0 µmol) and base (0.05-0.10 mmol) into each vial of the HTE plate.
- Liquid Handling: A liquid handling robot uses positive displacement tips to add stock solutions of aryl halide substrate (0.05 mmol in 0.5 mL solvent), coupling partner, and internal standard (durene or hexamethylbenzene for GC analysis).
- Inert Atmosphere: The entire plate is transferred to a glovebox or purged with an integrated N₂/Ar manifold for 15 minutes.
- Initiating Reaction: The plate is sealed with a PTFE/silicone mat and heated with agitation in a pre-heated multi-well aluminum block reactor at the target temperature (e.g., 80°C, 600 rpm).
- Quenching: After a defined period (e.g., 18h), the plate is transferred to an automated liquid handler which adds a standardized quenching solution (e.g., 100 µL of 1M HCl in EtOAc for base-sensitive reactions).
Protocol 2.2: High-Throughput Analytical Workflow
Objective: To quantitatively analyze the crude reaction mixtures from Protocol 2.1.
- Sample Preparation: An automated liquid handler performs a 1:10 dilution of each quenched reaction into a designated analysis plate using a compatible solvent (e.g., MeCN for UPLC-MS).
- UPLC-MS Analysis:
- Column: C18 reversed-phase, 2.1 x 30 mm, 1.7 µm particles.
- Gradient: 5% to 95% MeCN in H₂O (with 0.1% formic acid) over 1.2 minutes.
- Detection: UV Diode Array (210-400 nm) and Electrospray Mass Spectrometry (positive/negative mode switching).
- Data Processing: Chromatograms are automatically integrated. Yield is determined via:
- Internal Standard Method: Peak area of product vs. internal standard, calibrated with a standard curve.
- UV/ELSD Response Factor: Using estimated relative response factors.
- MS Signal: For rapid qualitative assessment only.
Visualizing the Integrated HTE Data Cycle
The efficiency of modern synthesis relies on a closed feedback loop between design, execution, and analysis.
Diagram Title: The Closed-Loop HTE Data Cycle
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents and Materials for HTE Synthesis
| Item | Function in HTE | Key Consideration |
|---|---|---|
| Pre-weighed Catalyst/ Ligand Plates | Provides exact, pre-dosed catalyst and ligand aliquots in each well of a microplate. | Eliminates manual weighing, ensures accuracy, and dramatically increases setup speed. |
| Automated Liquid Handlers (e.g., Hamilton, Beckman) | Precisely dispenses µL to mL volumes of solvents, substrates, and quenching agents. | Critical for reproducibility and enabling 24/7 operation. Integration with scheduling software is essential. |
| HTE Micro-Reactor Blocks (24-384 wells) | Parallelized, temperature-controlled reaction vessels with magnetic stirring. | Must be chemically resistant, have minimal well-to-well crosstalk, and compatible with automated sealers. |
| UPLC-MS Systems with Autosamplers | Provides rapid, information-rich analysis (mass spec, UV) of crude reaction mixtures. | Fast cycle time (<2 min/sample), robust to matrix effects, and software must enable batch processing. |
| Laboratory Information Management System (LIMS) / ELN | Central repository for structured experimental data, linking design parameters to analytical outcomes. | Must have APIs for robotic control and data ingestion; enables searchability and machine learning. |
| Design of Experiments (DOE) Software | Statistically plans reaction arrays to maximize information gain per experiment. | Moves beyond "one-variable-at-a-time" to efficiently map chemical space and discover interactions. |
Signaling Pathways in Catalysis: A Data-Driven Deconvolution
Understanding reaction outcomes requires mapping the complex network of catalytic and parasitic pathways.
Diagram Title: Key Pathways in a Model Cross-Coupling Catalytic Cycle
The core challenge of speed and data in modern synthesis is being met through the integration of deterministic automation, high-frequency analytical feedback, and intelligent data architecture. Within HTE batch reactor design, success is contingent on treating data as a primary product of the experimental process. The subsequent application of machine learning to these rich datasets promises to transform the iterative "make-test" cycle into a predictive, knowledge-generating engine, fundamentally accelerating the discovery and optimization of new chemical matter for drug development and beyond.
High-Throughput Experimentation (HTE) has fundamentally transformed organic synthesis research within drug discovery and development. Traditionally, HTE was synonymous with parallel synthesis—the rapid, simultaneous execution of numerous reactions under varied conditions, typically in microtiter plates. This approach excelled at surveying chemical space for initial hits. However, the contemporary thesis for modern HTE batch reactor design frames it as a paradigm of Integrated Experimentation. This evolved model integrates parallel synthesis with real-time analytics, automated data feedback loops, and intelligent decision-making algorithms. The core objective is no longer merely to run many reactions, but to generate maximal, actionable knowledge per unit volume and time, accelerating the development of robust, scalable synthetic routes.
Core Principles of Integrated HTE
Integrated HTE is characterized by three synergistic pillars:
- Systematic Reaction Parameter Investigation: Moving beyond one-variable-at-a-time (OVAT) to Design of Experiments (DoE) to explore multidimensional parameter spaces (e.g., solvent, ligand, base, concentration, temperature) and identify interactions.
- In-Line/On-Line Analytics: Direct integration of analytical techniques (e.g., HPLC, UPLC-MS, FTIR, NMR) with reactor blocks for real-time or rapid-quench reaction monitoring, transforming qualitative observations into quantitative kinetic and mechanistic data.
- Closed-Loop Workflows: Automation of the entire cycle from experiment design and execution to analysis, data interpretation, and the formulation of subsequent experimental hypotheses, often guided by machine learning (ML) models.
Modern HTE Batch Reactor Architecture
The physical manifestation of integrated HTE is a sophisticated batch reactor platform. A representative workflow is depicted below.
Diagram 1: Closed-loop integrated HTE workflow
Key Hardware Components & Specifications
Modern systems feature modular blocks of miniaturized batch reactors (typically 0.2-5 mL volume) with individual temperature and stirring control. Crucially, they are coupled directly to analytical instrument inlets.
Table 1: Specifications of a Representative Integrated HTE Platform
| Component | Typical Specification | Function in Integrated HTE |
|---|---|---|
| Reactor Format | 24-, 48-, or 96-well glass or metal blocks | Enables parallel reaction execution with controlled environment. |
| Temperature Range | -20°C to 150°C (with Peltier/fluid jacket) | Explores kinetic and thermodynamic regimes safely. |
| Agitation | Individual magnetic stirring (>500 rpm) | Ensures proper mixing and mass transfer in microliter volumes. |
| Atmosphere Control | Inert gas manifold (N₂, Ar) | Essential for air/moisture sensitive chemistries (organometallics). |
| Liquid Handling | Automated dispenser (µL precision) | Enables high-precision reagent addition for reproducibility. |
| Sampling Interface | Robotic syringe or solid-core sampler | Transfers aliquots directly to integrated LC/MS for analysis. |
Experimental Protocols for Integrated HTE
This section provides a detailed methodology for a classic HTE investigation: Palladium-Catalyzed Cross-Coupling Condition Screening.
Protocol: DoE-Driven Suzuki-Miyaura Coupling Screening
Objective: To identify optimal ligand, base, and solvent combinations for the coupling of an aryl bromide with an aryl boronic acid.
Materials: (See "The Scientist's Toolkit" below). Pre-Experiment Planning (DoE): Utilize a fractional factorial or Taguchi design to select 24-48 unique conditions from a matrix of:
- Ligands (8): SPhos, XPhos, RuPhos, t-BuXPhos, XantPhos, DPEPhos, P(2-furyl)₃, None.
- Bases (4): K₂CO₃, Cs₂CO₃, K₃PO₄, t-BuONa.
- Solvents (6): 1,4-Dioxane, Toluene, DMF, THF, EtOH/H₂O (1:1), MeCN/H₂O (1:1).
Procedure:
- Reactor Block Preparation: Under an inert atmosphere, load the HTE reactor block with clean, dry vials.
- Stock Solution Preparation: Prepare standardized stock solutions (0.1 M in appropriate solvent) of the aryl bromide substrate, boronic acid, and each candidate ligand.
- Automated Dispensing: a. Using liquid handling, dispense specified volumes of substrate, boronic acid, and ligand stocks into each vial according to the DoE layout. b. Dispense the assigned solvent to bring all vials to a uniform volume.
- Reaction Initiation: Using the dispensing robot, add a standardized aliquot of a Pd precursor (e.g., Pd(OAc)₂ or Pd₂(dba)₃ stock solution) to each vial, followed by the assigned base (solid or solution). Start agitation (900 rpm) immediately.
- Temperature Control: Set the block to the target temperature (e.g., 80°C) and start the reaction timer.
- Automated Sampling & Analysis: a. At predetermined timepoints (e.g., 1h, 4h, 18h), the robotic sampler withdraws a precise aliquot (e.g., 10 µL) from each vial. b. The sample is automatically diluted in a quenching solvent (e.g., MeOH with an internal standard) and injected into the integrated UPLC-MS. c. The UPLC-MS method quantifies the depletion of starting material and formation of product via UV and MS detection.
- Data Processing: Analytical software automatically integrates peaks and calculates conversion and yield (using internal standard calibration). Results are compiled into a structured data table.
Table 2: Example HTE Screening Data Output (Abridged)
| Well # | Ligand | Base | Solvent | Conv. (%) | Yield (%) | Purity (AUC %) |
|---|---|---|---|---|---|---|
| A1 | SPhos | K₂CO₃ | 1,4-Dioxane | 99 | 92 | 98 |
| A2 | XPhos | Cs₂CO₃ | Toluene | 95 | 88 | 97 |
| A3 | RuPhos | K₃PO₄ | DMF | 85 | 80 | 96 |
| B1 | None | t-BuONa | EtOH/H₂O | 10 | 5 | 90 |
| ... | ... | ... | ... | ... | ... | ... |
Data Integration & The Knowledge Feedback Loop
The raw data from Table 2 is processed to build a predictive model. The relationships in this feedback system are critical.
Diagram 2: HTE data-driven ML feedback loop
This loop transforms the platform from a screening tool into a discovery engine. The model can predict outcomes for untested conditions and guide the design of focused, informative follow-up experiments, such as kinetic profiling of the top conditions.
The Scientist's Toolkit: Essential Reagent Solutions
Table 3: Key Research Reagent Solutions for Integrated HTE
| Reagent/Material | Function & Rationale |
|---|---|
| Modular Ligand Kits | Pre-weighed, stabilized libraries of common phosphine and N-heterocyclic carbene (NHC) ligands in sealed vials. Enables rapid, consistent addition without manual weighing. |
| Pd/Transition Metal Precursor Stocks | Standardized solutions (e.g., 0.05 M in THF or toluene) of catalysts like Pd₂(dba)₃, Pd(OAc)₂, Ni(COD)₂. Critical for reproducible initiation of cross-coupling reactions. |
| Automated Base Dispenser | System for dispensing solid bases (carbonates, phosphates) or concentrated base solutions accurately into microliter-scale reactions. |
| Deuterated Solvent Spikes with Internal Standard | Used for rapid NMR analysis post-quench. Contains a known concentration of a standard (e.g., CH₂Br₂) for quantitative yield determination without purification. |
| Integrated LC/MS Vials with Pre-filled Quench Solvent | Vials located in the autosampler containing a known volume of quenching solvent (e.g., MeCN) and internal standard. The robotic sampler injects the reaction aliquot directly into this vial for immediate dilution and analysis. |
Within the broader thesis on High-Throughput Experimentation (HTE) batch reactor design for organic synthesis research, the platform's efficacy hinges on the seamless integration of its core physical components. This guide details the technical specifications, selection criteria, and operational protocols for the four pillars of a modern HTE batch reactor system: reaction vessels, heating/cooling, stirring, and automation. The convergence of these elements enables the rapid, parallel, and reproducible execution of chemical reactions, accelerating discovery in pharmaceutical and materials research.
Reaction Vessels: The Foundation
Vessels must be chemically resistant, withstand pressure/temperature cycles, and facilitate miniaturization.
Key Materials & Configurations:
- Glass Vials (e.g., HPLC, scintillation): For ambient pressure reactions. Often used with PTFE/silicone septa for sealing.
- PTFE/Silicone Inserts: Placed within standard wellplates, offering excellent chemical resistance for small-scale (0.5-2 mL) reactions.
- Stainless Steel Reactor Blocks: Machined blocks with individual reactor wells (e.g., 6, 24, or 48 reactors). Enable high-pressure/temperature (e.g., up to 200°C and 20 bar) reactions. Often combined with PTFE or glass inserts for chemical isolation.
Quantitative Comparison of Common Vessel Types
| Vessel Type | Typical Volume Range | Max Temp (°C) | Max Pressure (bar) | Key Material(s) | Primary Use Case |
|---|---|---|---|---|---|
| Glass Via + Septa | 1-20 mL | 150 (short term) | ~1 (sealed) | Borosilicate Glass, PTFE/Silicone | Ambient pressure HTE, stock solutions. |
| Wellplate with PTFE Insert | 0.5-2 mL | 120 | ~3 | Polypropylene/PTFE | Low-pressure, parallel synthesis & screening. |
| Modular SS Reactor (with insert) | 1-10 mL | 200 | 20-30 | Stainless Steel, PTFE/Glass | High-pressure/temperature catalysis, gas-liquid reactions. |
| Single SS Autoclave | 5-100 mL | 300 | 100+ | Stainless Steel, Inconel | Process scouting, extreme condition reactions. |
Heating & Cooling Systems
Precise, uniform, and rapid thermal control is non-negotiable for kinetic consistency across parallel experiments.
Methods:
- Conductive Heating/Cooling: Aluminum or copper blocks with embedded Peltier elements or cartridge heaters. Provides excellent uniformity for blocks holding multiple vials.
- Convective Heating: Forced air ovens (e.g., GC ovens). Slower ramp rates but can accommodate irregular geometries.
- Indirect Fluid Circulation: Heated/cooled fluid (e.g., silicon oil) circulated through channels in the reactor block. Offers high stability and wide temperature range (-80°C to +200°C).
Protocol: Standardized Temperature Equilibration Test
- Objective: Verify thermal uniformity across all reactor positions.
- Methodology:
- Fill all reactor vessels with 2 mL of a high-boiling point silicone oil.
- Insert calibrated thermocouples into at least 4 corner vessels and one center vessel.
- Set the heating block to a target temperature (e.g., 100°C).
- Monitor and log the temperature of each sensor until all reach steady state (±1°C of setpoint for 5 minutes).
- Record the time to reach steady state and the maximum observed temperature deviation between vessels.
- Acceptance Criterion: Maximum inter-vessel temperature deviation ≤ 2.0°C at steady state.
Agitation & Stirring
Effective mixing is critical for mass transfer, especially in heterogeneous or multiphase reactions.
Primary Technologies:
- Magnetic Stirring: Individual micro-stir bars driven by a rotating magnet array beneath the reactor block. Simplicity and scalability are key advantages.
- Orbital Shaking: The entire reactor block is agitated in an orbital path. Effective for mixing in small volumes without internal stir bars.
- Vortex Mixing: Periodic high-intensity vortexing. Often used in combination with other methods to re-suspend solids.
Stirring Method Selection Guide
| Stirring Method | Typical Speed Range | Suitability for Solids | Suitability for Gases | Scalability (to # of reactors) | Power Consumption |
|---|---|---|---|---|---|
| Individual Magnetic | 200-1200 rpm | High (with appropriate stir bar) | Moderate (dependent on baffling) | High (up to 96+) | Low-Medium |
| Orbital Shaking | 300-1500 rpm | Low-Moderate | Low | High (entire block) | Medium |
| Overhead Agitation | 50-1000 rpm | Very High | Very High (with gas entrainment) | Low (complex mechanics) | High |
Automation & Control Systems
Automation integrates components, enforces protocols, and enables data-rich experimentation.
Core Automated Functions:
- Liquid Handling: Robotics for reagent addition, sampling, and quenching.
- Gas Handling: Precensor pressure regulators and solenoid valves for gas charging/purging.
- Process Control: Software-controlled PID loops for temperature, pressure, and stirring speed.
- In-line Analytics: Integration with probes (FTIR, Raman) or automated sampling to HPLC/GC/MS.
Experimental Protocol: Automated Catalyst Screening Workflow
- Vessel Preparation: Robotic arm dispenses substrate solution (1.0 mL) into 24 PTFE-lined SS reactors.
- Catalyst/Additive Addition: A liquid handler adds a library of catalyst ligands (variable, 0.5-5 mol%) from stock solutions to individual wells.
- Inert Atmosphere: The reactor block is sealed, and a sequence of vacuum/N2 purges (3 cycles) is executed.
- Reaction Initiation: A common catalyst precursor solution is added simultaneously via a multi-port injector.
- Process Control: The system ramps to set temperature (e.g., 80°C) with stirring at 750 rpm for 18 hours.
- Automated Quenching: At t=18h, the block is cooled to 10°C, and a quenching agent (e.g., 0.1 mL acetic acid) is added to each well.
- Sampling: An automated sampler withdraws 10 µL from each well, dilutes it, and injects it into an HPLC for analysis.
Visualization: HTE Batch Reactor System Workflow
Title: Automated HTE Reaction Workflow Cycle
The Scientist's Toolkit: Essential Research Reagent Solutions
| Item | Function & Importance in HTE |
|---|---|
| PTFE/Silicone Septa | Provides a resealable, chemically inert barrier for vials, allowing for needle insertion for sampling or gas introduction without breaking atmosphere. |
| Micro Stir Bars (1-5 mm) | Enables efficient mixing in small volumes (0.2-5 mL). Coated in PTFE or glass for chemical inertness. Critical for solid suspension and gas dissolution. |
| Deuterated Solvents in Ampoules | Pre-packaged, anhydrous solvents for rapid preparation of NMR samples directly from reaction aliquots, essential for reaction monitoring and yield determination. |
| Internal Standard Solutions | Pre-mixed, precise solutions (e.g., 0.1M mesitylene in d8-THF) for automated addition to quenched samples, enabling accurate quantitative analysis by GC-FID or HPLC. |
| Catalyst/ Ligand Stock Solutions | Pre-weighed, standardized solutions in inert atmosphere vials. Enables rapid, accurate, and reproducible dispensing of small, catalytic quantities via liquid handling robots. |
| Pressure-Sensitive Indicator Caps | Specialized caps that change color if a safe pressure threshold is exceeded, providing a visual safety check for arrays of parallel pressure reactors. |
Within the broader thesis on High-Throughput Experimentation (HTE) batch reactor design for organic synthesis research, the selection of reactor materials is paramount. The core objective is to ensure chemical compatibility, maintain reaction integrity, prevent contamination, and guarantee reactor longevity under diverse and aggressive conditions. This guide details the critical material considerations for constructing or selecting batch reactors used in parallel synthesis, route scouting, and catalyst screening for pharmaceutical research.
Core Material Classes and Their Properties
The following tables summarize quantitative data on common reactor materials. All data is compiled from current manufacturer specifications and technical literature.
Table 1: Metallic Reactor Materials for HTE
| Material | Typical Alloy/Grade | Max Continuous Temp (°C) | Key Chemical Resistance | Primary Limitations | Common HTE Use Case |
|---|---|---|---|---|---|
| Stainless Steel | 316/316L | 900 | Good resistance to oxidizing acids, alkalis, organic solvents. | Poor against halides (Cl-, F-), reducing acids (HCl, HBr), pitting corrosion. | High-pressure/temp reactions with non-halogenated reagents. |
| Hastelloy | C-276 | 1090 | Excellent resistance to strong oxidizing & reducing agents, wet Cl₂, mixed acids. | High cost, machining difficulty. | Reactions involving HCl, H₂SO₄, halogenated species at high T/P. |
| Inconel | 625 | 980 | High strength, excellent oxidation resistance, good vs. organic acids. | Attacked by hot concentrated acids (H₂SO₄, HCl). | High-temperature oxidations, supercritical fluid reactions. |
| Titanium | Grade 2 | 300 (in air) | Outstanding resistance to chlorides, seawater, oxidizing acids. | Reactive with pure O₂, fluorine, anhydrous strong acids. | Chloride-mediated reactions, metal-catalyzed cross-couplings. |
Table 2: Non-Metallic & Lining Materials for HTE
| Material | Form/Type | Max Continuous Temp (°C) | Key Chemical Resistance | Primary Limitations | Common HTE Use Case |
|---|---|---|---|---|---|
| PTFE (Teflon) | Liner or Coating | 260 | Near-universal chemical inertness; resists all common solvents and acids/bases. | Low mechanical strength, permeability to small molecules, "creeps" under load. | Universal liner for vials/reactors, especially for harsh halogen chemistry. |
| PFA | Liner | 260 | Similar to PTFE, with better transparency and form stability. | Higher cost than PTFE, still permeable. | Liners for visual monitoring of reactions. |
| Glass (Borosilicate) | Reactor Insert | 500 (short term) | Excellent inertness to organics, acids (except HF, hot H₃PO₄), no metal leaching. | Brittle, attacked by strong alkalis and hydrofluoric acid. | Standard for small-scale HTE blocks, optical monitoring compatibility. |
| Ceramic (Alumina) | Monolithic or Coating | 1500 | Extreme wear resistance, inert to most molten chemicals and solvents. | Brittle, complex fabrication, thermal shock sensitivity. | Specialized high-temperature solid-phase or molten salt synthesis. |
Experimental Protocols for Material Compatibility Testing
Protocol 3.1: Static Immersion Test for Chemical Resistance
Objective: To quantitatively assess the mass change and visual degradation of candidate reactor materials upon exposure to key process reagents.
Methodology:
- Sample Preparation: Machine material coupons (e.g., 20mm x 10mm x 2mm). Polish to a uniform surface finish (e.g., 600 grit). Clean ultrasonically in acetone and isopropanol, then dry and weigh to 0.1 mg precision (initial mass, Mᵢ).
- Test Solution Preparation: Prepare 20 mL of test medium in a sealed vial. Options include: a) Simulated reaction mixture (solvent, substrates, catalysts, byproducts). b) Concentrated reagent (e.g., 12M HCl, 50% NaOH, 30% H₂O₂). c) Post-reaction crude mixture.
- Immersion: Immerse coupon in solution, ensuring no contact with other metals. Seal vessel. Use PTFE-lined caps.
- Conditioning: Place vessels in an oven or thermal block at the target process temperature (e.g., 80°C, 150°C) for a defined period (e.g., 24, 168, 1000 hours).
- Analysis:
- Post-Exposure Mass: Remove coupon, rinse with appropriate solvent, dry, and weigh (Mf). Calculate mass change: ΔM% = [(Mf - Mᵢ) / Mᵢ] x 100.
- Visual/Microscopic Inspection: Document pitting, cracking, discoloration, or etching using optical microscopy.
- Surface Analysis (Optional): Use SEM-EDS to detect surface composition changes and elemental leaching.
Protocol 3.2: High-Pressure Autoclave Stress Test
Objective: To evaluate material performance under combined chemical, thermal, and pressure stress, simulating actual reactor conditions.
Methodology:
- Reactor Setup: Install candidate material as a removable liner, insert, or fabricate a mini-autoclave from the material.
- Reaction Charge: Load with a challenging, representative reaction mixture (e.g., a Buchwald-Hartwig amination involving bases and polar solvents).
- Pressurization & Heating: Seal reactor, pressurize with inert gas (N₂) to target pressure (e.g., 10 bar). Heat with stirring to target temperature (e.g., 150°C) for a defined period (e.g., 72 hours).
- Cooling & Analysis: Cool, slowly vent pressure, and open reactor.
- Analyze Reaction Mixture: Use ICP-MS to quantify metal leaching from reactor walls into the product mixture.
- Inspect Reactor: Examine for signs of stress corrosion cracking, blistering (in coatings), or permanent deformation.
Decision Workflow and System Diagrams
Material Selection Logic for HTE Reactors
Material Compatibility Testing Workflow
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents & Materials for Compatibility Studies
| Item/Reagent | Function in Compatibility Testing | Typical Supplier/Example |
|---|---|---|
| Corrosive Test Solutions | Simulate aggressive reaction environments. | 37% HCl, 48% HBr, 70% HNO₃, 50% NaOH, 30% H₂O₂ (Sigma-Aldrich, Fisher). |
| Deuterated Solvents for NMR | Analyze reaction mixture post-exposure for decomposition products or leached material complexes. | DMSO-d₆, CDCl₃, D₂O (Cambridge Isotope Labs). |
| ICP-MS Standard Solutions | Calibrate ICP-MS for precise quantification of metal ion leaching (Cr, Ni, Mo, Fe, Ti). | Multi-element standard, 10 ppm in 2% HNO₃ (Inorganic Ventures). |
| PTFE/PFA Microvials & Liners | Provide inert containment for screening tests and as a control baseline. | 2 mL & 8 mL vials with screw caps (ChemGlass, vialab). |
| High-Temperature Epoxy | For sealing test coupons or constructing mock reactor assemblies. | TorrSeal (Varian), Aremco high-temp adhesives. |
| Non-Metallic Sampling Tools | Prevent contamination when handling test mixtures for analysis. | PTFE-coated forceps, polypropylene pipettes (VWR, Cole-Parmer). |
This whitepaper delineates the integration of High-Throughput Experimentation (HTE) batch reactor design with informatics workflows to accelerate organic synthesis research, particularly in pharmaceutical development. The data-centric paradigm treats experimental data as the primary, immutable asset, around which reactor design, execution, and analysis are orchestrated. This guide provides a technical framework for implementing this approach, complete with protocols, data standards, and visualization tools.
Traditional synthesis research often treats data collection as a secondary outcome of physical experimentation. The data-centric paradigm inverts this model. Reactor design—from micro-scale multi-well plates to parallel pressure vessels—is dictated by the requirements of downstream data generation, capture, and computational analysis. This linkage creates a closed-loop system where informatics workflows suggest experiments, and experimental outcomes refine predictive models.
Core Principles of Data-Centric HTE Reactor Design
An effective data-centric HTE system for organic synthesis rests on four pillars:
- Parameterization & Digitization: All reactor properties (volume, material, geometry, heating/cooling rate, stirring type) and process variables (T, P, time, concentration) must be defined in machine-readable formats (e.g., JSON schemas, XML).
- Inherent Data Capture: Sensors and analytical tools (inline FTIR, HPLC, GC) are integrated not as add-ons but as fundamental design components, generating structured, time-stamped data.
- Standardized Experimental Description: The use of standardized ontologies (e.g., RXNO for reactions, ChEBI for chemicals) to describe experiments enables universal data interoperability.
- Informatics-First Workflow: The experimental lifecycle is managed within a digital platform (Electronic Lab Notebook - ELN, Laboratory Information Management System - LIMS) from inception, linking hypothesis, procedure, raw data, and analysis.
Informatics Workflow Architecture
The workflow is a cyclic process of planning, execution, and learning.
Diagram Title: Data-Centric HTE Workflow Cycle
Key Experimental Protocols & Methodologies
This section details a standard protocol for a Suzuki-Miyaura cross-coupling HTE screen, a common reaction in drug discovery.
Protocol: HTE Screening for Suzuki-Miyaura Coupling
Objective: To rapidly identify optimal ligand and base combinations for a novel aryl chloride substrate.
Materials: See The Scientist's Toolkit below. Reactor Platform: 24-well glass-coated microtiter plate with magnetic stirring, housed in a modular parallel reactor block capable of inert atmosphere control and heating to 150°C.
Procedure:
- Digital Template Creation: In the ELN, a reaction template is created using a SMILES string for the novel aryl chloride and boronic acid. Variables are defined:
Ligand(list of 8 ligands),Base(list of 3 bases),Solvent(fixed: 1,4-dioxane/water 4:1). - Automated Liquid Handling: A liquid handler prepares stock solutions of substrates, catalyst (Pd source), ligands, and bases.
- Well Dispensing:
- Each well receives 1.5 mL of solvent mixture.
- Aryl chloride (0.1 mmol, 0.05 M final conc.) and boronic acid (0.12 mmol) are dispensed.
- Pd source (1 mol% Pd) and ligand (2 mol%) are added according to the combinatorial matrix.
- Base (2.0 equiv) is added last.
- Reaction Execution: The plate is sealed, placed under N₂ atmosphere, and heated at 80°C for 16 hours with constant stirring (750 rpm).
- Automated Quenching & Analysis: The plate is cooled to 25°C. An aliquot from each well is automatically diluted and analyzed by UPLC-MS.
- Data Processing: UPLC-MS data is automatically processed: peak integration yields conversion (aryl chloride consumption) and yield (via internal standard). Data is pushed to the data lake with metadata linking to the digital experiment ID.
Data Output & Tabulation
Typical results from such a screen are summarized below.
Table 1: Representative HTE Screen Results for Suzuki-Miyaura Coupling
| Well | Ligand Code | Base | Conversion (%) | UPLC Yield (%) | Purity (Area %) | Notes |
|---|---|---|---|---|---|---|
| A1 | SPhos | K₃PO₄ | 99 | 95 | 98 | Optimal condition |
| A2 | SPhos | Cs₂CO₃ | 99 | 93 | 97 | High yield |
| A3 | SPhos | t-BuONa | 15 | 10 | 85 | Low conversion |
| B1 | XPhos | K₃PO₄ | 99 | 90 | 96 | Good yield |
| B2 | XPhos | Cs₂CO₃ | 99 | 92 | 97 | Good yield |
| B3 | XPhos | t-BuONa | 40 | 35 | 90 | Moderate conversion |
| C1 | BippyPhos | K₃PO₄ | 5 | <2 | 70 | Ineffective |
| ... | ... | ... | ... | ... | ... | ... |
Table 2: Aggregate Statistical Analysis of Screen
| Metric | Value | Calculation Basis |
|---|---|---|
| Reactions Run | 24 | 8 ligands x 3 bases |
| Mean Yield | 68.2% | Average of UPLC Yield column |
| Yield Std Dev | ± 31.5% | Population standard deviation |
| High-Yielding Hits | 8 | Reactions with Yield ≥ 85% |
| Success Rate | 33.3% | (High-Yielding Hits / Total) * 100 |
| Optimal Condition | SPhos / K₃PO₄ | Highest yield & purity |
The Scientist's Toolkit: Essential Reagents & Materials
| Item | Function in HTE Organic Synthesis | Key Considerations for Data-Centric Use |
|---|---|---|
| Pd-Precursors (e.g., Pd(OAc)₂, Pd(dba)₂) | Catalytic source of Palladium for cross-couplings. | Stock solution concentration must be precisely logged in ELN for accurate molarity calculations. |
| Ligand Library (Buchwald, etc.) | Modifies catalyst activity & selectivity. | SMILES strings for each ligand must be stored in a searchable, digital chemical inventory. |
| Diverse Base Set (Carbonates, Phosphates, Alkoxides) | Scavenges acid byproduct, impacts rate. | Physical properties (pKa, solubility) should be included as metadata. |
| Deuterated Solvents for NMR | For reaction monitoring & yield determination. | Lot-specific residual proton signals should be documented to aid automated NMR analysis. |
| Internal Standard (e.g., 1,3,5-Trimethoxybenzene) | For quantitative GC/LC analysis. | Must be inert and elute separately from all reaction components; its exact concentration is a critical data point. |
| Anhydrous Solvents in Sure/Seal Bottles | Ensure reproducibility of air/moisture-sensitive reactions. | Bottle opening and usage should be tracked in inventory system to gauge solvent quality over time. |
| Chemically Resistant Microtiter Plates | High-density reaction vessels for parallel synthesis. | Plate geometry and material properties (thermal conductivity, adsorption) are key design parameters for heat/mass transfer models. |
Data Flow & System Integration
The integration of physical hardware and digital systems is critical. The following diagram maps the logical flow of data from reactor to repository.
Diagram Title: Logical Data Flow from Reactor to Database
The data-centric paradigm fundamentally re-engineers the process of organic synthesis research. By explicitly linking HTE reactor design decisions to the needs of informatics workflows—emphasizing standardization, inherent data capture, and digital continuity—research organizations can dramatically increase the velocity, reproducibility, and machine-learnability of their experimental programs. This approach transforms the laboratory from a data-generating facility into a component of a integrated, self-optimizing discovery engine.
Implementing HTE Batch Reactors: From Setup to Synthesis Workflows
The modern pursuit of molecular discovery in organic synthesis, particularly for drug development, demands rapid and efficient exploration of chemical space. This whitepaper details the critical upstream components of a High-Throughput Experimentation (HTE) workflow, framed within a broader thesis on designing batch reactor systems for organic synthesis research. The integration of meticulous reaction planning, rational plate layout, and precise reagent preparation directly dictates the success, reproducibility, and data density of HTE campaigns conducted in parallel batch reactors (e.g., 24-, 48-, 96-, or 384-well plates). This guide provides a technical framework for researchers and scientists to standardize and optimize these foundational steps.
Reaction Planning and Design of Experiment (DoE)
Effective HTE begins with strategic experimental design, moving beyond one-variable-at-a-time (OVAT) approaches to multivariate parameter screening.
Core Methodologies:
- Factor Selection: Identify key reaction variables (e.g., ligand, base, solvent, catalyst, temperature, concentration). Literature review and mechanistic hypotheses are crucial.
- DoE Strategy: Implement factorial or partial factorial designs (e.g., 2-level fractional factorial) to efficiently sample the parameter space. For exploring complex interactions, response surface methodologies (e.g., Central Composite Design) are employed post-initial screening.
- Control Wells: A minimum of 10% of wells should be designated for controls (e.g., positive/negative controls, internal standards for analytics). This is non-negotiable for robust data analysis.
Table 1: Common DoE Approaches for HTE Reaction Planning
| Design Type | Key Variables | Wells Required (Example) | Primary Use Case |
|---|---|---|---|
| Full Factorial | 4 factors, 2 levels | 16 | Comprehensive screening of all factor interactions when well count is not limiting. |
| Fractional Factorial | 6 factors, 2 levels | 16-32 | High-efficiency screening to identify main effects and some interactions. |
| Plackett-Burman | 11 factors, 2 levels | 12 | Very high-efficiency screening for main effects only. |
| Custom Array | Varies (e.g., 96 ligands) | 96+ | Focused screening of one parameter class (e.g., ligand library) against fixed background conditions. |
Diagram Title: HTE Reaction Planning Decision Workflow
Plate Layout and Logistics
The physical arrangement of experiments on a microtiter plate is a critical informatics challenge. A clear layout prevents handling errors and ensures data traceability.
Detailed Protocol for Layout Generation:
- Software Utilization: Use specialized software (e.g., Mosaic, ChemSpeed software, or custom Python/R scripts) or a master spreadsheet to map each unique reaction condition to a specific well coordinate (e.g., A01, B12).
- Replication: Include technical replicates (same condition in different wells) to assess intra-plate variability.
- Edge Effect Mitigation: Avoid placing critical test reactions on the perimeter wells, which are prone to evaporation. Use these wells for controls or blanks.
- Physical Template: Generate a printed "workmap" for the technician to follow during plate preparation. Color-coding for different reagent classes (e.g., catalysts, ligands) is highly recommended.
Table 2: Example 96-Well Plate Layout for a Solvent/Base Screen
| 1 (Control) | 2 (Solvent A) | 3 (Solvent B) | 4 (Solvent C) | 5 (Solvent D) | |
|---|---|---|---|---|---|
| A (Base 1) | Negative Ctrl | Condition 1 | Condition 2 | Condition 3 | Condition 4 |
| B (Base 2) | Positive Ctrl | Condition 5 | Condition 6 | Condition 7 | Condition 8 |
| C (Base 3) | Internal Std | Condition 9 | Condition 10 | Condition 11 | Condition 12 |
| D (No Base) | Solvent Blank | Condition 13 | Condition 14 | Condition 15 | Condition 16 |
Diagram Title: Plate Workflow from Preparation to Data
Reagent Preparation and Stock Solution Strategy
Consistency in reagent preparation is paramount. The use of master stock solutions is the standard for ensuring uniformity across an HTE plate.
Experimental Protocol for Stock Solution Preparation:
- Calculate Requirements: Based on the plate layout, calculate the total volume needed for each unique reagent, including a minimum 20% overage for liquid handling dead volume.
- Solvent Selection: Prepare all stock solutions in a dry, inert solvent compatible with the reaction (e.g., anhydrous DMSO, THF, toluene, DMF). Use calibrated balances and volumetric glassware.
- Concentration Standardization: Typical concentrations for catalysts/ligands are 5-50 mM. Substrate stock solutions are often prepared at 10x the target reaction concentration.
- Quality Control: Record lot numbers, preparation dates, and, if possible, perform NMR or titration assays on critical stock solutions (e.g., organometallic catalysts).
- Storage: Store solutions appropriately (e.g., under inert atmosphere, at low temperature) and note their stability lifetime.
Table 3: Example Stock Solution Table for a Cross-Coupling HTE
| Reagent | Role | Target Rxn Conc. (mM) | Stock Conc. (mM) | Stock Solvent | Stability |
|---|---|---|---|---|---|
| Pd(OAc)₂ | Catalyst | 0.5 | 5.0 | Anhydrous THF | 1 week, N₂, -20°C |
| Ligand Library | Ligand | 1.0 | 10.0 | Anhydrous DMSO | 1 month, N₂, RT |
| Cs₂CO₃ | Base | 2.0 | 40.0 (slurry) | Anhydrous DMF | 1 week, dried, RT |
| Aryl Halide | Substrate | 10.0 | 100.0 | Anhydrous Dioxane | Indefinite, N₂, RT |
The Scientist's Toolkit: Key Research Reagent Solutions
| Item / Solution | Function in HTE Workflow |
|---|---|
| Anhydrous DMSO | Universal polar solvent for preparing air/moisture-stable ligand and substrate stock solutions. High solubility for diverse organics. |
| Internal Standard Stock | A calibrated solution of a non-interfering compound (e.g., dodecane for GC, deuterated standard for NMR) added pre- or post-reaction for quantitative yield analysis. |
| Quenching Solution | A standardized reagent (e.g., aqueous HCl, silica gel suspension, scavenger resin) to uniformly stop reactions across the entire plate at a set time. |
| Calibrated Liquid Handler Tips | Disposable or washable tips for positive displacement or air displacement pipettors. Critical for accurate transfer of viscous or volatile solvents. |
| Inert Atmosphere Chamber/Glovebox | For preparation of oxygen/moisture-sensitive stock solutions and for charging reactors when reactions are highly air-sensitive. |
| Multi-Channel Pipettor or Liquid Handling Robot | Enables simultaneous addition of a single reagent (e.g., base, quench) to entire rows or columns of a microtiter plate, saving time and improving consistency. |
| Barcoded Vials & Plates | Provides unambiguous digital tracking of stock solutions and reaction plates from preparation through analysis, linking physical samples to digital data. |
| Digital Lab Notebook (ELN) Template | A pre-formatted digital template that captures all metadata: DoE structure, stock solution data, plate layout, and analytical results in a linked, searchable format. |
A robust HTE workflow for organic synthesis in batch reactors is built upon the interdependent pillars of statistically informed reaction planning, logical and error-minimized plate layout, and meticulous reagent preparation. Standardizing these upstream processes within the broader thesis of reactor design ensures that the high-quality data generated is a true reflection of chemical reactivity, not obscured by experimental artifact. This systematic approach enables researchers to confidently extract structure-activity or structure-reactivity relationships, accelerating the discovery and optimization of new synthetic methodologies and target molecules.
Within High-Throughput Experimentation (HTE) for organic synthesis research, the selection of reactor format is a fundamental design decision that influences throughput, data quality, operational flexibility, and cost. This guide provides an in-depth technical comparison of two dominant batch reactor formats: microtiter plates and individual miniature reactors (exemplified by carousel systems), framing the analysis within the context of optimizing HTE platforms for exploratory synthesis and reaction optimization.
Core System Architectures & Quantitative Comparison
Microtiter Plate Systems
These systems utilize standardized plates (e.g., 24, 48, 96, 384-well) as arrays of discrete, fixed-volume reaction vessels. Reactions are typically performed in parallel with shared environmental control (temperature, agitation). Liquid handling is accomplished via automated pipettors or dispensers.
Individual Miniature Reactor Carousels
These systems consist of an array of separate, sealed reactor vessels (0.5-5 mL) arranged on a carousel or cassette. Each reactor is an independent unit with individual control over stirring and often pressure. They are serviced by robotic arms for reagent addition and sampling.
Table 1: Quantitative System Comparison
| Parameter | Microtiter Plate Systems | Individual Miniature Reactor (Carousel) Systems |
|---|---|---|
| Typical Reaction Volume | 0.1 - 1.0 mL | 0.5 - 5.0 mL |
| Throughput (Experiments/run) | High (24 - 384+) | Medium (6 - 48) |
| Vessel Material | Polypropylene, Glass-coated | Borosilicate glass, PTFE, Stainless steel |
| Agitation | Orbital shaking (shared) | Individual magnetic stirring |
| Temperature Control | Single block for entire plate | Individual heating blocks or chambers |
| Pressure Tolerance | Atmospheric (or < 2 bar with seals) | High (commonly up to 20 bar) |
| Headspace Environment | Shared inert gas blanket | Individual vessel control (inert/vacuum) |
| Cross-Contamination Risk | Low to Moderate (splash) | Very Low (physically isolated) |
| Liquid Handling | Parallel dispensing (wells addressed in groups) | Serial addition via robotic needle |
| Capital Cost | Moderate | High |
Table 2: Suitability for Common Organic Synthesis Conditions
| Synthesis Condition | Microtiter Plate Performance | Carousel Reactor Performance |
|---|---|---|
| Air/Moisture Sensitive | Challenging; requires glovebox use | Excellent; individual sealed vessels |
| Elevated Temperature (>100°C) | Limited by plate material | Excellent (compatible with reflux) |
| Pressurized Gas (H₂, CO) | Not suitable | Excellent (standard feature) |
| Heterogeneous Catalysis | Poor mixing of solids | Excellent with magnetic stirring |
| Reaction Sampling | Difficult mid-run; usually endpoint | Straightforward via liquid handler |
| Scale-up Correlation | Poor (mixing/shaking artifact) | Good (mimics traditional flask) |
Detailed Experimental Protocols
Protocol 1: Parallel Suzuki-Miyaura Cross-Coupling in a 96-Well Plate
Objective: To screen 96 phosphine ligands against a single aryl halide and boronic acid pair.
Materials & Reagents: 96-well polypropylene plate with PTFE-lined silicone mat seal, stock solutions of Pd(OAc)₂ (in THF), aryl halide (in DMF), boronic acid (in DMF), ligand library (in DMF), base (Cs₂CO₃ in H₂O).
Methodology:
- Dispensing: Using an 8-channel pipettor or liquid handler, aliquot 100 µL of a 10 mM aryl halide solution into all 96 wells.
- Ligand Addition: Add 10 µL of each unique ligand solution (50 mM) to individual wells. Include control wells with no ligand.
- Catalyst Addition: Add 10 µL of Pd(OAc)₂ solution (5 mM) to all wells.
- Base Addition: Add 20 µL of Cs₂CO₃ solution (1.0 M) to all wells.
- Start Reaction: Add 60 µL of boronic acid solution (20 mM) to all wells to initiate coupling. Final volume: 200 µL.
- Seal & React: Apply a pressure-sensitive seal, mix on an orbital shaker (750 rpm), and heat in a convection oven at 80°C for 18 hours.
- Analysis: Cool plate, dilute all wells with 800 µL of MeOH, and analyze by UPLC-MS for conversion.
Protocol 2: Hydrogenation Screening in a Carousel Reactor System
Objective: To test 24 catalyst/substrate combinations under 5 bar H₂ pressure.
Materials & Reagents: 24× 4 mL glass vial reactors with magnetic stir bars and septa, catalyst library, substrate stock solution, liquid handling robot with gas-tight syringe.
Methodology:
- Vessel Charging: Robotic arm dispenses 2 mL of substrate solution (0.1 M in solvent) into each reactor.
- Catalyst Addition: Robotic arm adds a unique solid or liquid catalyst aliquot to each vessel.
- Sealing & Purging: Each reactor is sealed with a cap incorporating a needle port. The system evacuates the headspace of each vessel and purges with N₂ (3 cycles), then H₂ (3 cycles).
- Pressurization: The reactor block is pressurized to 5 bar with H₂.
- Reaction: Individual magnetic stirring (1000 rpm) and heating (e.g., 40°C) is initiated for 4 hours.
- Sampling & Analysis: At intervals, the robotic needle pierces a septum, samples 50 µL from a vessel, depressurizes it into a vial, dilutes, and injects into an online GC for analysis.
- Quenching: After the run, reactors are individually vented, and contents are sampled for final yield determination by NMR.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for HTE Reaction Screening
| Item | Function & Importance |
|---|---|
| Deuterated Solvents (DMSO-d6, CDCl3) | For direct reaction analysis by NMR in the screening plate/vessel. |
| Internal Standard Solutions | Precise volumetric addition (e.g., CH₂Br₂, 1,3,5-trimethoxybenzene) for quantitative GC/NMR analysis. |
| Pre-weighed Ligand/Catalyst Stock Plates | 96- or 384-well plates with pre-dispensed, nanomole-scale catalysts for rapid library execution. |
| Scavenger Resins | For post-reaction parallel workup to remove metals or excess reagents prior to analysis. |
| Colorimetric Quench Plates | Plates containing acid/base or dye indicators for rapid, visual assessment of reaction completion or pH. |
| PTFE/Silicone Septa Mats | Provide seal for microplates during heating and agitation, preventing evaporation and cross-contamination. |
| Gas Manifold Attachments | Enables purging of microplate wells with inert gas, improving compatibility with air-sensitive chemistry. |
HTE Platform Decision Workflow
Decision Workflow for Reactor Format Selection
Typical HTE Experimental Execution Flow
HTE Batch Reaction Execution Flow
The choice between microtiter plates and carousel-based miniature reactors is not one of superiority, but of application-specific suitability. For ultra-high-throughput screening of non-demanding reaction conditions where cost and experiment density are paramount, microtiter plates are indispensable. For advanced organic synthesis methodologies involving sensitive reagents, pressure, heterogeneous mixtures, or where individual vessel control and sampling are critical, carousel systems provide the necessary robustness and fidelity. The optimal HTE laboratory for organic synthesis research will strategically integrate both platforms, aligning the reactor format with the specific chemical challenge and stage of investigation.
Within the framework of High-Throughput Experimentation (HTE) batch reactor design for organic synthesis research, the precise handling of air- and moisture-sensitive compounds, as well as the management of pressure, is foundational. This guide details the technical protocols, materials, and engineering controls required to achieve reproducibility and safety in parallel screening environments common to modern drug development.
Core Principles and Engineering Controls
Inert Atmosphere Management
The primary goal is to exclude O₂ and H₂O to prevent undesired oxidation, hydrolysis, or quenching of reactive intermediates (e.g., organometallics, strong bases, pyrophoric reagents).
Key Quantitative Data: Table 1: Common Inert Gases and Their Purification Specifications
| Gas | Typical Purity (%) | Primary Impurity | Purification Method | Final H₂O (ppm) | Final O₂ (ppm) |
|---|---|---|---|---|---|
| Nitrogen (N₂) | 99.998 | O₂, H₂O | Dual-Column Gas Purifier | < 1 | < 1 |
| Argon (Ar) | 99.999 | N₂, H₂O | Hot Metal Getter (Cu) | < 0.1 | < 0.1 |
| Helium (He) | 99.999 | N₂, H₂O | Molecular Sieve Trap | < 1 | < 1 |
Experimental Protocol: Glovebox Operation and Reactor Charging
- System Preparation: Ensure the glovebox (maintained at < 1 ppm O₂ and H₂O) is under positive pressure. Log O₂ and moisture analyzer readings.
- Antechamber Purge: Place dried glassware and solid reagents in the antechamber. Execute a minimum of three purge-evacuate cycles with inert gas.
- Reagent Handling: Inside the main chamber, charge HTE reactor vials with solids using a microbalance. Seal vials with septa.
- Liquid Transfer: Use gastight syringes to transfer air-sensitive liquids (e.g., n-BuLi, TiCl₄) through the vial septum.
- Reactor Sealing: Secure reactor blocks with torque-limiting wrenches to ensure uniform sealing force (typically 10-15 N·m).
- Removal: Place the sealed reactor block into the antechamber and execute an evacuation-backfill cycle before removal.
Moisture Control
Water is a pervasive contaminant. Control spans from macroscopic reactor design to molecular-level reagent purification.
Key Quantitative Data: Table 2: Common Drying Agents and Solvent Specifications
| Drying Agent/Solvent | Residual H₂O (ppm) After Drying | Capacity | Reactivity Notes | Suitability for HTE |
|---|---|---|---|---|
| Molecular Sieves (3Å or 4Å) | < 10 | High | Non-reactive, requires activation | Excellent for solvent reservoirs |
| n-BuLi in Hexanes | N/A | N/A | Quenches H₂O, titrated for concentration | Critical reagent, used via syringe |
| Solvent Dispensing System (SDS) | < 50 (Karl Fischer) | N/A | In-line column, under N₂ pressure | Essential for parallel synthesis |
| P₂O₅ | < 5 | Very High | Violent reaction with H₂O, not for solvents | Used in desiccators |
Experimental Protocol: Solvent Drying and Titration of Air-Sensitive Reagents
- Solvent Drying: Reflux technical-grade solvent (e.g., THF) over sodium/benzophenone under N₂ until a deep blue/purple ketyl radical anion color persists. Distill under inert atmosphere directly into an SDS reservoir containing activated 3Å molecular sieves.
- Reagent Titration (e.g., n-BuLi): a. Prepare a 1.0 M solution of 2-butanol in dry xylene. b. Under N₂, add 1.0 mL of this solution to a flask containing 10 mL of dry hexane and 2-3 drops of 1,10-phenanthroline indicator. c. Titrate with the n-BuLi solution until the yellow indicator turns red. Calculate molarity: M_BuLi = (1.0 M * 1.0 mL) / (mL of BuLi used).
Pressure Management in HTE Reactors
HTE reactors must safely contain reactions that generate gas (e.g., H₂, CO₂) or are run under elevated pressure (e.g., hydrogenations, high-temperature transformations).
Key Quantitative Data: Table 3: HTE Reactor Pressure Ratings and Safety Parameters
| Reactor Vial Type | Max Working Pressure (psig) | Burst Pressure (psig) | Typical Seal Material | Temp. Range (°C) |
|---|---|---|---|---|
| Glass (Thick-walled) | 100 | 300 | PTFE-faced Silicone | -80 to 150 |
| Stainless Steel | 1500 | 4500 | Metal C-ring (Cr-Ni alloy) | -200 to 350 |
| PFA Liner in SS | 500 | 1500 | PTFE | -200 to 260 |
Experimental Protocol: Conducting a High-Pressure Hydrogenation in an HTE Batch Reactor
- Risk Assessment: Calculate theoretical H₂ consumption and final pressure. Ensure all components are rated for the maximum expected pressure.
- Reactor Charging: In a glovebox, weigh catalyst and substrate into a PFA liner. Add dry solvent via SDS.
- Sealing: Assemble the liner inside the steel reactor block. Tighten closure bolts in a cross-pattern to the manufacturer's specified torque.
- Pressurization: Connect the reactor block to a manifold. Perform three vacuum/H₂ purge cycles at 50 psig to remove N₂. Finally, pressurize with H₂ to the target pressure (e.g., 200 psig).
- Reaction & Monitoring: Place the block in a pre-heated stirring/heating station. Monitor pressure drop as an indicator of reaction progress.
- Safe Venting: After cooling to room temperature in an ice bath, vent the H₂ slowly in a fume hood. Confirm pressure is atmospheric before disassembly.
The Scientist's Toolkit: Essential Materials for HTE of Sensitive Chemistry
Table 4: Research Reagent Solutions & Essential Materials
| Item | Function & Explanation |
|---|---|
| Inert Atmosphere Glovebox | Maintains an environment with <1 ppm O₂/H₂O for weighing solids, assembling reactions, and storing sensitive reagents. |
| Solvent Drying/Dispensing System (SDS) | Integrated system that stores solvents over drying agents and dispenses them via syringe or cannula under positive inert gas pressure, preventing atmospheric contamination. |
| Gastight Syringes (Hamilton) | Precision syringes with PTFE seals for accurate transfer of air- and moisture-sensitive liquids without ingress of atmosphere. |
| Automatic Reagent Titrator | Instrument for precise, repeatable determination of concentrations of reactive reagents (e.g., organolithiums, Grignards) via colorimetric or potentiometric methods. |
| High-Pressure HTE Reactor Block | Parallel array reactor (e.g., 6-24 wells) made of stainless steel or alloy, capable of sealing individual vials and withstanding simultaneous heating, stirring, and pressurization. |
| Torque-Limiting Wrench/Driver | Ensures uniform and manufacturer-specified sealing force on reactor vial caps, critical for preventing leaks and ensuring consistent reaction volumes. |
| Moisture/Oxygen Analyzer | In-line or handheld sensors (e.g., electrochemical, laser-based) for real-time monitoring of glovebox atmosphere or gas stream purity. |
| Pressure Transducer/Data Logger | Electronic sensors connected to each reactor vial to monitor pressure in real-time, providing kinetic data for gas-consuming/gas-evolving reactions. |
| PFA (Perfluoroalkoxy) Liners | Chemically inert, disposable liners for stainless steel reactor vials, preventing catalyst poisoning and simplifying cleaning, especially for metal-catalyzed reactions. |
| Self-Sealing Septa (PTFE/Silicone) | Pre-slit septa for reactor vials that allow needle penetration for liquid addition/pressure relief while maintaining a resealable, pressure-rated barrier. |
Visualized Workflows and Relationships
Diagram Title: Workflow for HTE Reactor Setup Under Inert Conditions
Diagram Title: Impact of Atmosphere Control on HTE Reaction Outcomes
Integrating In-Line and At-Line Analytical Techniques (ReactIR, GC/MS, UHPLC)
The drive towards accelerated process development in pharmaceutical organic synthesis necessitates a paradigm shift in laboratory experimentation. High-Throughput Experimentation (HTE) in batch reactor design generates vast amounts of empirical data, but its true potential is unlocked only when coupled with sophisticated, real-time analytics. This whitepaper posits that the strategic integration of in-line (e.g., ReactIR) and at-line (e.g., GC/MS, UHPLC) analytical techniques forms the core analytical engine for modern HTE platforms. This integration transforms HTE from a screening tool into a deep knowledge-generation system, enabling precise kinetic profiling, mechanistic elucidation, and rapid failure analysis within complex experimental matrices.
Core Analytical Techniques: Functions & Synergies
In-line Analysis with ReactIR
Function: Provides continuous, non-invasive monitoring of reaction progress in real-time by measuring vibrational spectra (typically in the mid-IR region) via attenuated total reflectance (ATR) probes immersed directly in the reaction medium. Primary HTE Application: Tracking the consumption of key reagents (e.g., carbonyl species, azides) and the formation of products or intermediates with distinct IR fingerprints. Enables immediate feedback on reaction initiation, completion, or stalling.
At-line Analysis with Gas Chromatography/Mass Spectrometry (GC/MS)
Function: Provides separated, definitive identification and quantification of volatile and semi-volatile components. Samples are automatically or manually extracted from the reactor and injected. Primary HTE Application: Profiling complex reaction mixtures, identifying low-abundance byproducts or degradants, and confirming the identity of unknown components via spectral libraries. Essential for side-reaction mapping.
At-line Analysis with Ultra-High Performance Liquid Chromatography (UHPLC)
Function: Offers high-resolution separation, identification, and quantification of non-volatile and thermally labile compounds, typically with UV/Vis or mass spectrometric detection. Primary HTE Application: Accurate yield determination (via external calibration), assessing purity, and quantifying polar impurities. The workhorse for quantitative analysis in medicinal chemistry and process research.
Synergistic Data Integration
The power lies in the data hierarchy: ReactIR offers continuous kinetic data (when did something happen?), GC/MS provides qualitative structural insight (what unknown formed?), and UHPLC delivers quantitative analytical rigor (how much of each component exists?). Together, they create a complete reaction narrative.
Quantitative Comparison of Techniques
The following table summarizes the key characteristics of each technique in the context of an HTE batch reactor workflow.
Table 1: Technical Comparison of Integrated Analytical Techniques for HTE
| Parameter | ReactIR (In-line) | GC/MS (At-line) | UHPLC (At-line) |
|---|---|---|---|
| Measurement Type | Continuous, real-time | Discrete, post-sampling | Discrete, post-sampling |
| Sample Preparation | None (direct immersion) | Often required (dilution, derivatization) | Required (filtration, dilution) |
| Analysis Time per Sample | Seconds (continuous scan) | 5-30 minutes | 3-15 minutes |
| Primary Information | Kinetic profiles, functional group presence | Volatile component identity, purity | Quantification, non-volatile purity, yield |
| Key Strength in HTE | Real-time feedback, pathway elucidation | Unknown identification, byproduct detection | High-precision quantification, regulatory readiness |
| Key Limitation | Sensitivity to matrix effects, requires IR-active species | Limited to volatile/ derivatizable compounds | Method development time, longer cycle time |
| Typical Data Output | Concentration vs. time trend | Chromatogram with mass spectra | Chromatogram with peak area/height |
Experimental Protocols for Integrated Workflow
Protocol A: Real-Time Kinetic Study with At-Line Verification
Aim: To determine the kinetics of a catalytic coupling reaction and identify major byproducts.
Materials: HTE batch reactor block (e.g., 24-vessel), ReactIR with multiplexed ATR probes, automated liquid handler, GC/MS system, UHPLC-MS system.
Procedure:
- Reactor Setup: Charge each vessel with substrate, catalyst, and solvent under inert atmosphere using an automated liquid handler.
- In-line Monitoring: Start ReactIR data acquisition (e.g., 1 scan/min) for all vessels simultaneously upon initiation of the reaction (e.g., by reagent addition via robot).
- Scheduled At-line Sampling: Program the liquid handler to withdraw aliquots (e.g., 100 µL) from designated vessels at key timepoints (t=5, 30, 60, 120 min) based on ReactIR trend indications.
- Sample Processing:
- For GC/MS: Dilute aliquot 1:10 in appropriate solvent, transfer to GC vial.
- For UHPLC: Dilute aliquot 1:20 in methanol, filter through a 0.2 µm PTFE membrane into an HPLC vial.
- Sequential Analysis: Queue processed samples for GC/MS (for byproduct screening) and UHPLC (for precise yield calculation using a calibrated method).
- Data Correlation: Overlay ReactIR concentration trends (e.g., substrate disappearance) with UHPLC-derived yield data at matched timepoints to validate the in-line model. Use GC/MS identifications to annotate unexpected features in the IR or UHPLC data.
Protocol B: Reaction Failure Analysis and Optimization
Aim: To diagnose the cause of low yield in a specific HTE reaction condition.
Procedure:
- Flagging: Identify outlier vessel(s) from HTE block via real-time ReactIR trace showing incomplete conversion or unexpected spectral features.
- Immediate At-line Analysis: Prioritize the failed reaction vessel for expedited at-line analysis.
- GC/MS First-Pass: Rapidly analyze a crude sample by GC/MS to identify any volatile byproducts or residual starting materials not clearly visible by IR.
- UHPLC Quantification: Perform a precise UHPLC analysis to quantify remaining starting material and major product(s).
- Triangulation:
- ReactIR shows an intermediate plateau → GC/MS identifies a stable intermediate → UHPLC confirms its accumulation.
- ReactIR shows no reaction → GC/MS/UHPLC confirm starting material intact → Suggests catalyst/initiator failure.
- ReactIR shows rapid decay of product peak → UHPLC confirms low final yield, GC/MS identifies degradation products.
Visualization of Integrated Workflow & Data Flow
Diagram 1: Integrated Analytical Workflow for HTE
Diagram 2: Data Synergy Creating a Reaction Narrative
The Scientist's Toolkit: Essential Research Reagent Solutions & Materials
Table 2: Key Reagents and Materials for Integrated HTE Analytics
| Item | Function in Integrated Workflow | Key Consideration for HTE |
|---|---|---|
| ReactIR SiComp ATR Probe | In-line immersion probe for mid-IR spectroscopy; chemically resistant with silicon crystal. | Compatibility with a wide range of solvents and temperatures (typically -80°C to 200°C). |
| Multiplexer (MUX) | Enables a single ReactIR spectrometer to sequentially monitor multiple (e.g., 4-8) reactor vessels. | Critical for cost-effective scaling of in-line monitoring across an HTE block. |
| Automated Liquid Handler | Performs precise reagent addition and timed aliquot withdrawals from HTE vessels. | Must be integrated with reactor control software and compatible with vial/plate formats for at-line instruments. |
| GC/MS with Autosampler | Provides automated, high-throughput analysis of volatile components. | Fast GC columns and methods (≤10 min) are essential to keep pace with HTE output. |
| UHPLC-MS with Autosampler | Provides automated, high-resolution quantitative analysis. | Use of sub-2µm particle columns for fast separations. Method scalability is crucial. |
| Standardized Solvent Kits | Pre-mixed, degassed solvents for UHPLC mobile phases and sample dilution. | Ensures consistency and reduces preparation time across hundreds of HTE samples. |
| Internal Standards (IS) | Compounds added to all analysis samples to correct for injection variability (GC/MS, UHPLC). | Must be inert, stable, and elute separately from all reaction components. |
| Calibration Standard Sets | Pre-prepared solutions of known concentration for quantifying key analytes via UHPLC. | Enables rapid generation of calibration curves for high-precision yield determination. |
| 0.2 µm PTFE Syringe Filters | For crude reaction mixture filtration prior to UHPLC analysis to protect the column. | Essential for removing particulates and catalysts (e.g., Pd on carbon) from samples. |
This whitepaper details advanced methodologies for catalyst screening, reaction optimization, and solvent selection within the framework of High-Throughput Experimentation (HTE) batch reactor design for organic synthesis. The integration of HTE enables the rapid, parallel investigation of chemical space, which is critical for accelerating drug discovery and process chemistry research. This guide provides actionable protocols and data analysis strategies for implementing these techniques in a modern laboratory setting.
Catalyst Screening via HTE
Catalyst screening is a primary application of HTE, allowing for the evaluation of dozens to hundreds of potential catalysts in parallel to identify lead candidates for a given transformation.
Experimental Protocol for Catalytic Cross-Coupling Screening
Objective: Identify the most effective Pd-based catalyst for a model Suzuki-Miyaura cross-coupling.
Materials: Aryl halide (1.0 equiv), boronic acid (1.5 equiv), base (2.0 equiv), solvent (THF/H2O mixture), library of Pd catalysts (5 mol%).
HTE Workflow:
- Stock Solution Preparation: Prepare separate stock solutions of substrate, boronic acid, and base in the chosen solvent.
- Liquid Handling: Using an automated liquid handler, aliquot the substrate solution into individual wells of a 96-well microtiter plate designed for batch reactions.
- Catalyst Dispensing: Add a different Pd catalyst precursor from a library (e.g., Pd(PPh3)4, Pd(dppf)Cl2, Pd(OAc)2 with various ligands) to each well.
- Reagent Addition: Add boronic acid and base solutions to all wells.
- Reaction Execution: Seal the plate and heat in a modular HTE batch reactor block at 80°C for 18 hours with agitation.
- Quenching & Analysis: Cool plate, quench with a standard solution, and analyze conversion/yield via UPLC-MS equipped with an autosampler.
Representative Catalyst Screening Data
Table 1: Yield Data for Suzuki-Miyaura Catalysis Screen
| Catalyst/Ligand System | Pd Source | Ligand | Average Yield (%) | Notes |
|---|---|---|---|---|
| SPhos Pd G3 | Pd(allyl)Cl | SPhos | 98 | Most robust, high conversion |
| XPhos Pd G2 | Pd(allyl)Cl | XPhos | 95 | Excellent for steric hindrance |
| Pd(dppf)Cl2 | Pd(dppf)Cl2 | (dppf) | 88 | Good for electron-poor substrates |
| Pd(PPh3)4 | Pd(0) tetrahedral | (PPh3)4 | 75 | Moderate, baseline performance |
| Pd(OAc)2 / tBuXPhos | Pd(OAc)2 | tBuXPhos | 82 | Sensitive to oxygen/water |
Reaction Condition Optimization
Following catalyst identification, systematic optimization of reaction variables (temperature, time, concentration, stoichiometry) is performed using Design of Experiment (DoE) principles.
DoE Protocol for Condition Optimization
Objective: Optimize temperature, time, and catalyst loading for the identified lead catalyst (SPhos Pd G3).
Design: A 3-factor, 2-level full factorial design (8 experiments + 3 center points).
Factors & Levels:
- Factor A (Temperature): 60°C (-1), 80°C (0), 100°C (+1)
- Factor B (Time): 4h (-1), 12h (0), 20h (+1)
- Factor C (Catalyst Loading): 1 mol% (-1), 3 mol% (0), 5 mol% (+1)
Protocol:
- DoE Software: Use software (e.g., JMP, Design-Expert) to generate the experimental matrix.
- HTE Setup: Prepare master stocks. Use automated liquid handling to set up the 11 distinct reaction conditions in a 24-well HTE reactor block.
- Execution & Analysis: Run reactions in parallel, quench, and determine yield via UPLC-MS. Input yield data into DoE software for model generation.
Optimization Results
Table 2: DoE Matrix and Yield Results for Condition Optimization
| Expt. | Temp (°C) | Time (h) | Catalyst (mol%) | Yield (%) |
|---|---|---|---|---|
| 1 | 60 | 4 | 1.0 | 45 |
| 2 | 100 | 4 | 1.0 | 78 |
| 3 | 60 | 20 | 1.0 | 82 |
| 4 | 100 | 20 | 1.0 | 96 |
| 5 | 60 | 4 | 5.0 | 65 |
| 6 | 100 | 4 | 5.0 | 92 |
| 7 | 60 | 20 | 5.0 | 94 |
| 8 | 100 | 20 | 5.0 | 99 |
| 9-11 (CP) | 80 | 12 | 3.0 | 95±2 |
Conclusion: The model identified 100°C, 20h, and 5 mol% catalyst as optimal for maximum yield (>99%). A cost-saving alternative of 80°C, 12h, 3 mol% provides 95% yield.
Systematic Solvent Selection
Solvent choice profoundly impacts reaction rate, mechanism, and selectivity. HTE enables efficient mapping of solvent effects.
Protocol for Solvent Screen
Objective: Determine the optimal solvent for a nucleophilic aromatic substitution (SNAr).
Library: A diverse set of 12 solvents spanning different polarity, proticity, and coordinating ability.
Procedure:
- Plate Setup: In a 24-well HTE plate, dispense a constant volume of each pure solvent.
- Reagent Addition: Using automation, add identical aliquots of substrate, nucleophile, and base stocks to each well.
- Reaction: Seal and heat the plate at a fixed temperature (e.g., 50°C) for a fixed time (6h) with shaking.
- Analysis: Quench and analyze for conversion and byproduct formation via UPLC-MS.
Solvent Screen Data
Table 3: Solvent Effect on Model SNAr Reaction Yield
| Solvent | Class | Dielectric Constant (ε) | Yield (%) | Key Observation |
|---|---|---|---|---|
| DMF | Dipolar Aprotic | 38.3 | 98 | Optimal yield, clean reaction |
| DMSO | Dipolar Aprotic | 46.7 | 95 | Slightly slower |
| NMP | Dipolar Aprotic | 32.2 | 97 | Comparable to DMF |
| Acetonitrile | Dipolar Aprotic | 37.5 | 85 | Moderate yield |
| 1,4-Dioxane | Ether | 2.2 | 15 | Very low conversion |
| THF | Ether | 7.5 | 22 | Low conversion |
| MeOH | Protic | 32.7 | 40 | Side product formation |
| Toluene | Non-polar | 2.4 | <5 | No reaction |
Conclusion: Dipolar aprotic solvents (DMF, DMSO, NMP) are optimal, aligning with the SNAr mechanism's requirement to stabilize the anionic transition state.
The Scientist's Toolkit: Research Reagent Solutions
Table 4: Essential Materials for HTE in Organic Synthesis
| Item/Category | Function & Rationale |
|---|---|
| Modular HTE Batch Reactors (e.g., ChemSpeed, Unchained Labs) | Parallel, automated platforms for conducting reactions under controlled inert atmosphere, pressure, and temperature. |
| Automated Liquid Handler | Enables precise, reproducible dispensing of reagents, catalysts, and solvents to microtiter plates. |
| UPLC-MS with Autosampler | Provides rapid, quantitative analysis of reaction outcomes (conversion, yield, purity). Essential for high-throughput data generation. |
| 96-/384-Well Microtiter Plates (Glass- or Polymer-based) | Standardized reaction vessels compatible with automation and HTE reactor blocks. |
| Precatalyst & Ligand Libraries | Commercially available sets (e.g., Pd PEPPSI, Buchwald ligands, organocatalysts) for rapid catalyst screening. |
| Diverse Solvent Libraries | Curated sets covering a wide range of polarity, proticity, and coordinating ability for systematic solvent screening. |
| DoE Software (JMP, Design-Expert) | Used to design efficient experimental matrices and perform statistical analysis on multivariate data. |
| Laboratory Information Management System (LIMS) | Tracks samples, experimental parameters, and analytical data, ensuring data integrity and reproducibility. |
An effective HTE campaign integrates these steps sequentially: 1) Broad catalyst screening identifies a lead system; 2) DoE-driven optimization fine-tunes conditions for performance and robustness; and 3) Systematic solvent selection confirms or discovers the optimal reaction medium. This iterative, data-rich approach, enabled by modern HTE batch reactor design, dramatically accelerates the development of efficient synthetic routes in pharmaceutical research.
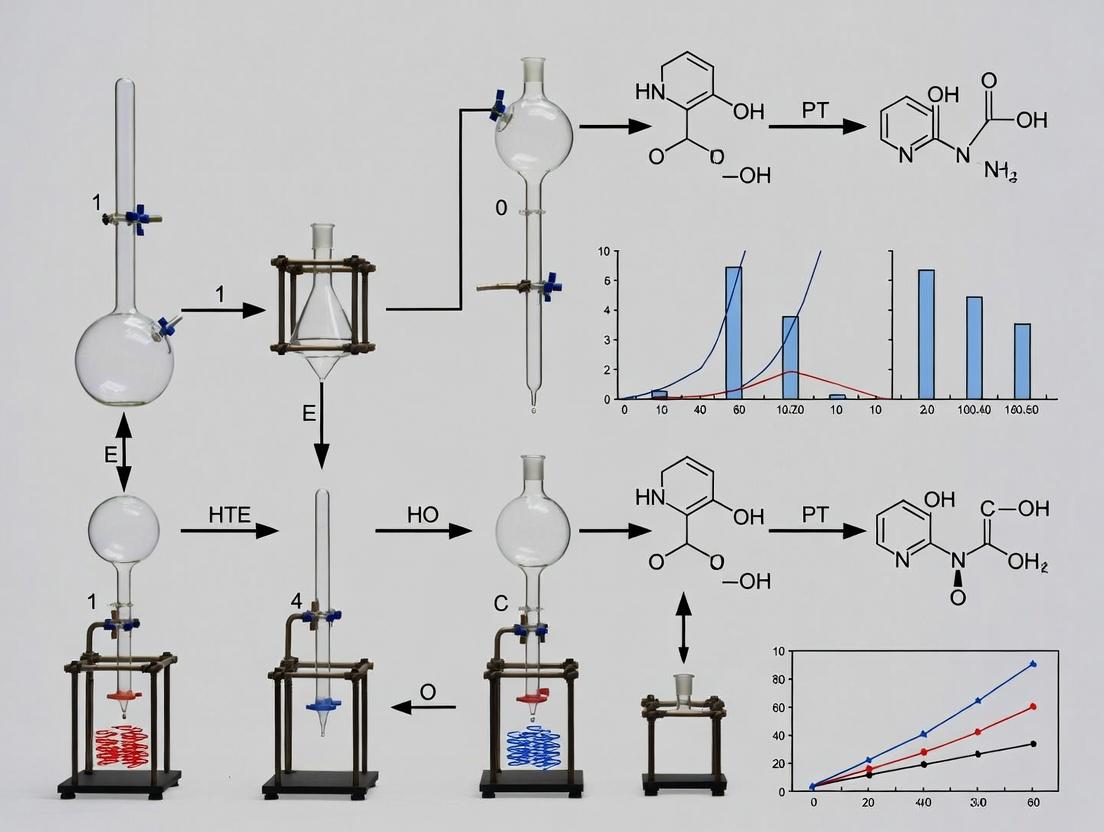
Solving Common HTE Reactor Challenges and Maximizing Data Quality
Addressing Evaporation and Solvent Loss in Small-Volume Reactions
Within the paradigm of High-Throughput Experimentation (HTE) for organic synthesis, the drive towards miniaturization—using reaction volumes in the microliter to low milliliter range—is fundamental for accelerating drug discovery. This scale enables rapid screening of vast reaction spaces while conserving precious substrates and reagents. However, a critical, often underestimated challenge arises: evaporative solvent loss. In the context of HTE batch reactor design, this is not a mere inconvenience but a systematic error source. Uncontrolled evaporation alters reagent concentrations, shifts reaction equilibria, increases viscosity, and can lead to complete reaction failure. This whitepaper provides an in-depth technical guide to mitigating this issue, ensuring data integrity and reproducibility in small-volume organic synthesis.
The Physics of Evaporation in Miniaturized Systems
Evaporative loss is governed by the equation for evaporative flux, J (mol m⁻² s⁻¹): J = (P_sat - P_partial) * √(M / (2πRT)) Where P_sat is the solvent's vapor pressure at temperature T, P_partial is its partial pressure in the headspace, M is molar mass, R is the gas constant.
In small-volume reactors, the surface-area-to-volume (SA:V) ratio is drastically increased. A 100 µL droplet in a 1 mL vial has a far greater relative exposure surface than 10 mL in a 20 mL vial. Consequently, even minor percentage losses represent catastrophic concentration changes. Key factors include:
- Solvent Volatility: Governed by vapor pressure.
- Temperature: Exponential impact on P_sat.
- Headspace Volume & Agitation: Larger headspace and agitation increase mass transfer.
- Reactor Seal Integrity: The primary engineering control point.
Table 1: Calculated Concentration Increase Due to Solvent Evaporation
| Initial Volume (µL) | Solvent Lost (µL) | % Volume Lost | Final Concentration Factor | Impact on 1 mM Starting Material |
|---|---|---|---|---|
| 1000 | 50 | 5% | 1.05x | 1.05 mM |
| 200 | 20 | 10% | 1.11x | 1.11 mM |
| 50 | 7.5 | 15% | 1.18x | 1.18 mM |
| 20 | 5 | 25% | 1.33x | 1.33 mM |
Table 2: Vapor Pressure of Common Organic Solvents at 25°C
| Solvent | Boiling Point (°C) | Vapor Pressure (mmHg) | Relative Evaporation Rate (BuAc=1) |
|---|---|---|---|
| Diethyl Ether | 34.6 | 537 | ~8.3 |
| Dichloromethane (DCM) | 39.8 | 435 | ~6.8 |
| Acetone | 56.1 | 231 | ~3.8 |
| Tetrahydrofuran (THF) | 66.0 | 162 | ~2.4 |
| Ethyl Acetate (EtOAc) | 77.1 | 73 | ~1.0 |
| Toluene | 110.6 | 28 | ~0.5 |
| N,N-Dimethylformamide (DMF) | 153.0 | 3.7 | ~0.06 |
| Dimethyl Sulfoxide (DMSO) | 189.0 | 0.42 | ~0.006 |
Experimental Protocols for Mitigation and Measurement
Protocol 1: Gravimetric Assessment of Vial Seal Integrity
Objective: Quantify evaporative loss from different sealed HTE reactor types. Materials: 96-well plate (0.5-2 mL/well) with sealing mat, 1.5 mL crimp vials with PTFE/silicone septa, 8 mL vials with screw caps, microbalance (±0.01 mg), DCM, DMF. Method:
- Tare each empty vessel.
- Precisely pipette 200 µL of test solvent (e.g., DCM and DMF) into selected wells/vials (n=5 per condition).
- Immediately apply the specified seal (mats crimped, caps torqued to 5 in-lbs).
- Weigh each sealed vessel and record as W₀.
- Place vessels on an HTE platform agitator at 500 rpm, 25°C, for 24 hours.
- Re-weigh vessels (Wₜ).
- Calculate: % Mass Loss = [(W₀ - Wₜ) / W₀(solvent)] * 100. Interpretation: Directly compares sealing technologies under operational conditions.
Protocol 2: Internal Standard Method forIn SituConcentration Monitoring
Objective: Monitor effective concentration change during a reaction. Materials: Reaction components, a non-volatile, chemically inert internal standard (IS) e.g., 1,3,5-trimethoxybenzene. Method:
- Prepare reaction master mix including the IS at a known molar ratio to the limiting reagent.
- Aliquot equal volumes into multiple identical reaction vessels.
- At designated time points (t=0, 2, 6, 24h), quench individual vessels.
- Analyze by quantitative methods (e.g., UPLC, GC).
- Calculate: Ratio of analyte peak area / IS peak area at each time point (Aₐₙₐₗyₜₑ/IS).
- Normalize all ratios to the t=0 ratio. A consistent increase over time indicates solvent evaporation concentrating all species.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Materials for Evaporation Control
| Item | Function & Rationale |
|---|---|
| PTFE/Silicone Laminated Septa Screw Caps | Provides a chemically inert, re-sealable barrier with low permeability to organic vapors. Essential for reliable vial-based HTE. |
| Pierceable, Heat-Sealing Foils & Mats | Enable sterile, gas-tight sealing of microplates. Aluminum-backed foils offer the best barrier; silicone/PTFE mats are for shorter durations. |
| Glass Inserts for Vials | Placing a small-volume reaction (e.g., 100 µL) inside a larger sealed vial (e.g., 2 mL) drastically reduces the effective SA:V ratio and creates a solvent-saturated headspace. |
| Non-Volatile, Inert Solvents (e.g., DMSO, DMF, NMP) | Using high-boiling solvents as the primary reaction medium intrinsically reduces vapor pressure-driven loss. |
| Seal Integrity Tester (e.g., Capacitive Sensor) | Instrument to non-destructively validate the seal on individual wells of a microplate before and after agitation. |
| Pre-hydrated Seals/Desiccant Packs | For reactions sensitive to atmospheric moisture, pre-equilibrating seals prevents evaporation into the seal, which can draw water out of desiccants. |
Optimized Sealing and Workflow Strategies for HTE
A systematic, tiered approach is required, integrating sealing technology with experimental design.
Diagram 1: HTE Sealing Strategy Decision Tree (92 chars)
Workflow Integration: This decision logic should be integrated into HTE protocol generation software. For critical screenings, Protocol 2 (Internal Standard) should be run in parallel on control wells to validate the chosen sealing strategy's efficacy over the entire campaign duration.
In HTE-driven organic synthesis, controlling evaporative solvent loss is not a peripheral consideration but a cornerstone of robust experimental design. By understanding the governing physics, quantitatively assessing the risk via tools like internal standards, and implementing a tiered strategy of sealing technologies, researchers can ensure that the valuable data generated in miniature reactors accurately reflects chemistry, not artifact. This rigor is essential for building reliable reaction databases that inform machine learning models and drive efficient drug development.
Ensuring Reproducible Mixing and Heat Transfer Across All Wells
Within the broader thesis on High-Throughput Experimentation (HTE) batch reactor design for organic synthesis, the central challenge is achieving true parallel reproducibility. For reaction discovery and optimization—especially in pharmaceutical research—the physical uniformity of mixing and heat transfer across all wells in a multi-reactor block is a prerequisite for meaningful data. This technical guide details the principles, validation protocols, and experimental methodologies required to ensure each well behaves as an identical, predictable chemical reactor.
Core Engineering Principles for Uniformity
Fluid Dynamics in Miniaturized Batch Reactors
Mixing in small-volume (0.5-5 mL) wells is dominated by viscous forces and interfacial phenomena, not inertia. Laminar flow conditions (Re < 100) are typical, making reproducible mixing highly dependent on impeller/vessel geometry and agitation frequency.
Fundamentals of Heat Transfer
Heat flow (Q) is governed by: Q = U * A * ΔT, where U is the overall heat transfer coefficient, A is the heat transfer area, and ΔT is the temperature gradient. In HTE blocks, variations in well-to-block contact, medium conduction, and local ambient conditions create thermal gradients that must be characterized and mitigated.
Table 1: Key Parameters Influencing Mixing & Heat Transfer Reproducibility
| Parameter | Impact on Mixing | Impact on Heat Transfer | Typical Target Tolerance |
|---|---|---|---|
| Agitation Speed (RPM) | Directly affects shear & flow. Primary mixing control. | Minor indirect effect via forced convection at interfaces. | ≤ ±2% CV across block |
| Well Liquid Volume | Affects vortex depth and gas-liquid interface. | Changes thermal mass and liquid-contact area ratio. | ≤ ±1% of set volume |
| Vessel Material & Wall Thickness | Affects wettability and fluid-wall interaction. | Major determinant of conductive resistance (1/U). | Consistent to ±0.1 mm |
| Block Material Thermal Conductivity | Negligible direct effect. | Primary path for heating/cooling. Aluminum (>200 W/m·K) preferred. | N/A |
| Impeller Geometry & Position | Critical for flow pattern reproducibility. | Affects internal convection uniformity. | Centered to ±0.5 mm |
Quantitative Characterization Protocols
Mixing Homogeneity Assessment: Dye Decolorization Kinetics
Objective: Quantify mixing efficiency and uniformity across wells by measuring the kinetics of a rapid, mixing-controlled chemical reaction.
Protocol:
- Reagent Preparation: Prepare a 1.0 mM solution of aqueous methylene blue. Prepare a 10% (w/v) solution of sodium dithionite (Na₂S₂O₄) in deoxygenated water.
- Experimental Setup: In all wells of the HTE block, pipette 2.0 mL of the methylene blue solution using a calibrated positive-displacement liquid handler.
- Initiation & Measurement: Using a simultaneous multi-channel pipettor, rapidly add 100 µL of the sodium dithionite solution to each well. Immediately initiate agitation at the target RPM. Monitor the decrease in absorbance at 664 nm for each well in real-time using a parallel photometric plate reader.
- Data Analysis: The decolorization follows pseudo-first-order kinetics. Plot ln(Absorbance) vs. Time for each well. The slope is the observed rate constant (k_obs), which is mixing-limited under these conditions.
- Uniformity Metric: Calculate the Coefficient of Variation (CV) of k_obs across all wells. A CV < 5% indicates acceptable mixing uniformity.
Table 2: Representative Dye Test Data for a 24-Well Block (Target RPM: 800)
| Well Position | k_obs (s⁻¹) | Time to 95% Completion (s) | Mixing Efficiency vs. Mean |
|---|---|---|---|
| A1 | 0.105 | 28.5 | 98.1% |
| A2 | 0.108 | 27.7 | 101.0% |
| B1 | 0.102 | 29.3 | 95.3% |
| ... | ... | ... | ... |
| D6 | 0.107 | 28.0 | 100.0% |
| Mean | 0.107 | 28.1 | 100% |
| CV | 2.1% | 2.3% | N/A |
Heat Transfer Uniformity Assessment: Thermal Calorimetry
Objective: Map the thermal response and steady-state uniformity across the reactor block.
Protocol:
- System Preparation: Fill all wells with 2.0 mL of a reference fluid (e.g., water or a common solvent like DMF).
- Instrumentation: Fit each vessel with a calibrated micro-thermocouple or resistance temperature detector (RTD) connected to a multi-channel data logger.
- Dynamic Ramp Test: Program the heating/cooling system for a defined ramp (e.g., 25°C to 80°C at 2°C/min). Record the temperature of every well at 5-second intervals.
- Steady-State Hold Test: At the target temperature (e.g., 80°C), hold for 30 minutes and log data.
- Data Analysis: For each well, calculate: a) Ramp Lag/Load (time difference to reach 50°C), b) Overshoot (max temp exceeded), and c) Steady-State Stability (mean ± SD during final 10 min hold).
Table 3: Thermal Performance Data for a 48-Well Block (Target: 80°C)
| Well Zone | Avg. Ramp Rate (°C/min) | Steady-State Mean Temp (°C) | Steady-State Std Dev (°C) | Max Observed ΔT from Setpoint |
|---|---|---|---|---|
| Center (Well Group 1) | 1.98 | 79.8 | 0.15 | -0.2 |
| Edge (Well Group 2) | 1.92 | 79.5 | 0.22 | -0.5 |
| Corner (Well Group 3) | 1.85 | 79.1 | 0.25 | -0.9 |
| Overall Block Mean | 1.92 | 79.5 | 0.21 | -0.5 |
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 4: Key Reagents & Materials for Reproducibility Validation
| Item | Function & Rationale |
|---|---|
| Calibrated Positive-Displacement Pipettor | Ensures identical liquid volumes in all wells, removing a major variable in thermal mass and mixing dynamics. |
| Methylene Blue / Sodium Dithionite System | Provides a rapid, colorimetric, mixing-sensitive chemical reaction for quantitative kinetic comparison across wells. |
| Traceable Micro-thermocouples (e.g., T-type) | Enables direct, simultaneous measurement of individual well temperatures with high accuracy for thermal mapping. |
| Standardized Viscosity Solution (e.g., 60% Glycerol) | Used to validate mixing under non-Newtonian or high-viscosity conditions relevant to real synthesis mixtures. |
| Infrared Thermal Imaging Camera | Allows for non-contact visualization of surface temperature gradients across the entire reactor block in real-time. |
| Reactor Well Pressure Monitors | Critical for reactions with gas evolution or under inert atmosphere; pressure can significantly impact mixing and boiling points. |
Workflow for System Validation & Experimental Design
Diagram Title: HTE Reactor Block Validation Workflow
Diagram Title: Causes and Effects of Poor Reproducibility
Advanced Mitigation Strategies
- Active Thermal Uniformity Control: Implement individual well heating via Peltier elements or resistive micro-heaters with PID feedback, rather than relying on a single block heater.
- Alternative Agitation: Consider orbital shaking or magnetic stirring with standardized stir bars for more consistent fluid dynamics than overhead stirring, especially for suspensions.
- Computational Fluid Dynamics (CFD) Modeling: Simulate fluid flow and heat transfer during reactor block design to predict and eliminate "cold spots" or "dead zones" prior to fabrication.
- Standardized Lidding & Insulation: Use thermally conductive, sealed lids to minimize evaporative cooling differences and ensure uniform headspace pressure.
Integrating the quantitative validation protocols and materials outlined in this guide transforms an HTE batch reactor system from a source of experimental noise into a foundation of reliable data. For drug development professionals, this reproducibility in mixing and heat transfer is not merely operational—it is the critical link that allows high-throughput data to accurately inform reaction mechanism understanding, scale-up predictions, and ultimately, the acceleration of synthetic route discovery.
Managing Exotherms and Ensuring Safety in High-Density Experiment Arrays
1. Introduction Within the broader thesis on High-Throughput Experimentation (HTE) batch reactor design for organic synthesis, managing thermal hazards is paramount. High-density arrays, where multiple reactions run in parallel under varying conditions, amplify the risk of exothermic runaway events. This guide details the principles and protocols for identifying, mitigating, and ensuring operational safety against such exotherms in dense reactor arrays.
2. Quantitative Risk Assessment: Calorimetric Data Primary screening via reaction calorimetry provides essential safety data. The following table summarizes key parameters for common organic synthesis reaction classes, as per current industry standards (2024-2025).
Table 1: Calorimetric Parameters for Common Reaction Classes
| Reaction Class | Typical ΔT_ad (°C) | Typical MTSR (°C) | TMR_ad (h) at 80°C | Severity Class (Stoessel) | Probability Class (Stoessel) |
|---|---|---|---|---|---|
| Nitration | 120-200 | 150-220 | <1 | High (4) | High (4) |
| Grignard | 80-150 | 110-180 | 2-8 | Medium (3) | Medium (3) |
| Epoxidation | 60-120 | 90-150 | 8-24 | Medium (2) | Low (2) |
| Esterification | 40-80 | 70-120 | >24 | Low (1) | Low (1) |
| Amide Coupling | 30-60 | 60-110 | >24 | Low (1) | Low (1) |
ΔT_ad: Adiabatic temperature rise; MTSR: Maximum Temperature of the Synthesis Reaction; TMR_ad: Time to Maximum Rate under adiabatic conditions.
3. Experimental Protocol: RC1e Reaction Calorimetry Screening Objective: Determine the heat flow and accumulation of a novel reaction prior to HTE array deployment. Materials: Mettler Toledo RC1e calorimeter, 1L glass reactor, dosing unit, thermocouples. Procedure:
- Charge substrate and solvent into the calibrated reactor. Set initial temperature (T0).
- Establish thermal baseline under stirring.
- Initiate reagent addition via automated dosing at a controlled rate (e.g., 1 mL/min).
- Record heat flow (Qr), temperature (Tp), and pressure in real-time.
- Calculate total reaction enthalpy (ΔHr), adiabatic temperature rise (ΔTad = ΔH_r / (m·Cp)), and MTSR.
- Perform a heat-wait-search (HWS) experiment to determine onset temperature (T_D24) for decomposition.
4. Safety-by-Design in HTE Array Configuration Based on calorimetric data, engineering controls are implemented at the array level.
Table 2: Array Engineering Controls Based on Risk Class
| Stoessel Risk Class | Max Wells per Heater Block | Recommended Well Volume (µL) | Mandatory Array Features | Quenching Protocol |
|---|---|---|---|---|
| High (4, 3) | ≤ 4 (isolated zones) | 100-500 | Independent Peltier cooling, pressure sensors per well, in-situ IR thermography | Automated cryogenic injector |
| Medium (2) | ≤ 16 | 500-1000 | Zonal temperature control, vibration stirring | Parallel liquid quench station |
| Low (1) | 24-96 | 1000-2000 | Block-level heating/cooling, magnetic stirring | Post-reaction plate handler |
5. Protocol: Parallel In-Situ Infrared Thermography for Hot-Spot Detection Objective: Visually identify and localize exothermic events in a 24-well HTE array in real-time. Materials: 24-well aluminum block reactor, FLIR A8580 MWIR camera (3-5 µm), IR-transparent sapphire window viewports, HTE robotic liquid handler. Procedure:
- Mount the IR camera at a fixed distance to image the entire array window.
- Calibrate camera emissivity for common reactor materials (Al, glass, PTFE).
- Initiate parallel reactions via rapid reagent dispensing.
- Acquire IR images at 5 Hz frequency.
- Process images to generate temperature maps and identify wells exceeding (T_p + 10°C).
- Trigger automated safety response (cooling or quenching) for identified hot-spots.
6. The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Reagents and Materials for Exothermic Risk Mitigation
| Item/Reagent | Function & Rationale |
|---|---|
| In-Situ FTIR Probes (ReactIR) | Monitors reagent consumption and intermediate formation in real-time, identifying kinetic instabilities. |
| Phase-Transfer Catalysts (e.g., Aliquat 336) | Facilitates reactions in biphasic systems at lower temperatures, reducing thermal load. |
| Solid-Supported Reagents (e.g., polymer-bound DCC) | Allows controlled, slow release of reactive species, minimizing heat accumulation. |
| Diluent Solvents (e.g., CPME, 2-MeTHF) | High heat capacity, low vapor pressure solvents act as thermal buffers. |
| Emergency Quench Solutions | Pre-loaded solutions (e.g., aqueous Na₂SO₃ for peroxides, acetic acid for organometallics) for rapid runaway suppression. |
| Sacrificial Base Scavengers | Polymer-bound sulfonic acids or amines to sequester excess reagents post-reaction safely. |
7. Logical Decision Workflow for Array Safety The following diagram outlines the decision-making pathway from reaction screening to safe array operation.
Diagram Title: HTE Safety Decision Workflow
8. Real-Time Safety Monitoring & Feedback System Architecture This diagram details the integrated monitoring and control system for a high-risk experiment array.
Diagram Title: Real-Time Safety Monitoring System
Strategies for Solid Handling and Heterogeneous Reaction Mixtures.
The push for accelerated discovery in organic synthesis, particularly within pharmaceutical research, demands efficient exploration of chemical space. High-Throughput Experimentation (HTE) in batch reactors is a cornerstone of this approach. However, the prevalence of solid reagents, catalysts, and by-products introduces significant complexity, turning effective handling of heterogeneous mixtures from a practical concern into a critical design variable for any HTE thesis. This guide details strategies to manage these challenges, ensuring reliability, reproducibility, and scalability in parallel reaction screening.
Fundamental Challenges in Heterogeneous HTE
Heterogeneity impacts every stage of an HTE workflow. Key challenges include:
- Dispersion and Contact: Ensuring consistent solid-liquid or solid-solid contact across dozens to hundreds of parallel reactors.
- Dosing Accuracy: Precise weighing and transfer of solid particulates, especially in sub-milligram scales.
- Agglomeration and Caking: Solids can aggregate, leading to reduced surface area and erratic reaction rates.
- Clogging: Solid handling can compromise liquid handling robotics and vial/well plate orifices.
- Sampling and Analysis: Withdrawing a representative aliquot from a slurry without phase separation is non-trivial.
- Scale-Up Disconnect: Reactions that perform well in a stirred HTE plate may fail in a kilo lab due to differing mixing dynamics.
Core Strategies and Methodologies
Physical Form Modification
The properties of the solid itself can be engineered for improved handling.
Protocol: Preparation of Polymer-Bound Reagents for Enhanced Dispersion
- Material: Weigh 1.0 g of the target reagent (e.g., an oxidant, reductant, or base).
- Immobilization: Dissolve the reagent and a compatible polymer scaffold (e.g., polystyrene, PEG) in a minimal volume of an appropriate solvent (e.g., DCM, THF).
- Precipitation: Rapidly add the solution to a large volume (50 mL) of a non-solvent (e.g., hexane, methanol) under vigorous stirring to co-precipitate the reagent onto the polymer.
- Isolation: Filter the resulting fine suspension and dry under vacuum overnight.
- Characterization: Determine loading (mmol/g) via elemental analysis or a standardized titration assay.
Advanced Agitation and Mixing
Conventional magnetic stirring is often inadequate for deep-well plates. Alternative strategies are required.
Protocol: Optimizing Mixing in a 96-Well HTE Plate
- Platform Selection: Use an HTE shaker/oscillator capable of orbital, linear, or figure-8 motion at frequencies up to 1500 rpm.
- Bead Addition: To each reaction well, add a single 3-4 mm diameter glass, PTFE, or stainless steel ball bearing.
- Parameter Screening: Set up a reaction known to be mixing-sensitive (e.g., a Pd-catalyzed cross-coupling with a solid base). Run identical reactions across the plate while varying the shaking frequency (e.g., 500, 750, 1000, 1250 rpm) and diameter (e.g., 1, 3, 5 mm).
- Analysis: Use UPLC conversion analysis to determine the minimum agitation threshold for consistent, high yield. This defines the baseline for all subsequent heterogeneous screens.
Precise Solid Dispensing
Accurate, parallel solid dosing is a major bottleneck. Solutions range from manual to fully automated.
Protocol: Manual Slurry Dispensing for Air-Sensitive Solids
- Slurry Preparation: In a nitrogen-filled glovebox, prepare a homogeneous slurry by combining the finely powdered solid (e.g., a metal catalyst) with a stable, inert solvent (e.g., mineral oil, dried Nujol) at a known concentration (e.g., 0.1 M).
- Syringe Use: Equip a gas-tight syringe with a wide-bore (e.g., 14-gauge) needle.
- Dispensing: While maintaining gentle magnetic stirring of the slurry reservoir, withdraw the required volume. Quickly dispense into the target reaction vials.
- Calibration: Establish a density correlation between slurry concentration and mass delivered per unit volume via gravimetric analysis.
In SituGeneration and Sequestration
Instead of handling problematic solids, they can be generated or trapped within the reaction medium.
Protocol: In Situ Acid Chloride Formation with By-Product Sequestration
- Reaction Setup: Charge a 2-dram vial with the carboxylic acid (1.0 mmol) and a polymer-bound base (e.g., PS-DMAP, 1.2 mmol).
- Activating Agent Addition: Add a solution of the chlorinating agent (e.g., oxalyl chloride, 1.05 mmol) in DCM (2 mL).
- Agitation: Cap and agitate the reaction on an HTE shaker (1000 rpm, 3 mm diameter) for 2 hours.
- Filtration: Use a filter plate or a simple pipette with a cotton plug to directly transfer the resulting in situ-generated acid chloride solution into a new vial containing the nucleophile (e.g., an amine). The solid by-products (HCl salts, polymer-bound reagents) are removed.
Quantitative Comparison of Agitation Methods
The choice of mixing strategy profoundly impacts reaction outcome in heterogeneous systems.
Table 1: Performance Comparison of Agitation Methods for a Model Suzuki-Miyaura Coupling with Solid K₃PO₄ Base
| Agitation Method | Hardware | Avg. Yield (%)* | Yield Std. Dev. (±%) | Practical Scale Limit | Risk of Cross-Contamination |
|---|---|---|---|---|---|
| Magnetic Stirring | Micro-stir bars | 45 | 25 | 1 mL | Low |
| Orbital Shaking | HTE shaker, loose beads | 78 | 12 | 5 mL | Medium |
| Vortex Mixing | Individual vortex mixer | 92 | 5 | 2 mL | High (if open) |
| Acoustic Mixing | Focused acoustics (e.g., EDC) | 95 | 3 | 1 mL | Very Low |
*Average of n=8 replicates per condition. Reaction: 4-bromotoluene (0.1 M), phenylboronic acid (1.2 eq.), Pd(dtbpf)Cl₂ (1 mol%), K₃PO₄ (2.0 eq. solid), 80°C, 18h.
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Materials for Heterogeneous HTE Workflows
| Item | Function & Rationale |
|---|---|
| Polymer-Bound Reagents (PS, PEG) | Immobilizes reagents/catalysts, simplifying filtration and purification, enabling use of excess without workup complications. |
| Molecular Sieves (3Å, 4Å) | Inexpensive, high-capacity solids for in situ water scavenging in moisture-sensitive reactions. |
| Dispensing Balls (Glass, PTFE, SS) | Provides intense, localized mixing in small volumes within well plates and vials under orbital shaking. |
| Filter Plates & SPE Cartridges | Enables rapid parallel solid-liquid separation directly in HTE blocks, crucial for quenching and sampling. |
| Slurry Dispensing Kits | Specialized syringes and reservoirs for handling suspensions of solids with robotic liquid handlers. |
| High-Density Solvents (Fluorous, Ionic Liquids) | Acts as a immiscible reaction medium or for sequestering catalysts, facilitating product isolation. |
| Scavenger Resins (e.g., QuadraPure) | Selectively removes excess reagents or by-products (e.g., metals, acids) post-reaction via filtration. |
Visualizing the HTE Workflow for Heterogeneous Systems
A systematic approach is vital for managing complexity and generating interpretable data.
Title: HTE Workflow for Heterogeneous Reaction Optimization
Title: Solid Handling via Reagent Immobilization
In conclusion, robust strategies for solid handling are not ancillary but central to the thesis of modern HTE-driven organic synthesis. By deliberately selecting from and integrating physical modification, engineered agitation, precise dispensing, and smart reaction design, researchers can transform heterogeneity from a source of irreproducibility into a controlled, even advantageous, parameter. This systematic approach ensures that promising leads identified in microtiter plates are grounded in scalable reality, accelerating the entire drug discovery pipeline.
This guide addresses a critical bottleneck in High-Throughput Experimentation (HTE) for organic synthesis: the imbalance between the number of parallel reactions executed in batch reactors and the downstream capacity to analyze the results. Within the broader thesis on HTE batch reactor design, optimizing this balance is paramount to realizing the promised acceleration of discovery in medicinal chemistry and drug development. A system with a 384-well reactor plate is futile if analytical throughput is limited to 10 samples per day.
The Core Throughput Challenge: Experiment vs. Analysis
The fundamental equation governing workflow efficiency is: Effective Throughput = min(Reaction Throughput, Analytical Throughput)
Reaction throughput in modern HTE has surged with automation, enabling hundreds to thousands of experiments per week. Analytical throughput, however, often lags due to longer run times, sample preparation bottlenecks, and data processing complexity.
Quantitative Comparison of Common Analytical Techniques
Data sourced from current literature and manufacturer specifications (2024).
Table 1: Throughput and Suitability of Key Analytical Techniques for HTE
| Analytical Technique | Avg. Time per Sample (min) | Max Samples per Day (8h) | Key Information Gained | Best for HTE Phase |
|---|---|---|---|---|
| UPLC-MS (Short Column) | 2.5 - 5 | 96 - 192 | Conversion, Identity, Purity | Primary Screening |
| SFC-MS | 1.5 - 3 | 160 - 320 | Chiral Separation, Purity | Enantioselective Screening |
| NMR (Flow, 96-well) | 5 - 10 | 48 - 96 | Structure Confirmation, Yield | Secondary Analysis |
| GC-MS | 3 - 7 | 68 - 160 | Volatile Compounds, Reaction Monitoring | Specific Reaction Classes |
| HPLC-UV (Rapid Grad.) | 4 - 8 | 60 - 120 | Purity, Conversion (UV-active) | Routine Quality Check |
| MS Flow Injection (FIA-MS) | 0.25 - 0.5 | 960 - 1920 | Presence/Absence of Product | Ultra-High-Throughput Triage |
Detailed Experimental Protocols for Balanced Workflows
Protocol: Tiered Analytical Triage for 384-Reaction Screening
Objective: Maximize information recovery from a large reaction array without analytical backlog. Materials: 384-well reactor block, automated liquid handler, UPLC-MS, FIA-MS system, 96-well plate deep-well dilution blocks.
Methodology:
- Reaction Execution: Perform reactions in 0.2-1 mL volume in 384-well plate. Quench uniformly via automated addition of standard solvent.
- Primary Triage (FIA-MS):
- Prepare a 1:100 dilution of each quenched reaction into a new 384-well plate using an automated liquid handler.
- Inject 5 µL from each well directly into the MS source via flow injection (no chromatography).
- Analysis: Use automated data processing to flag wells showing mass ion for desired product (M+/-). Expected throughput: ~1500 samples/day.
- Secondary Analysis (UPLC-MS) on Triage Hits:
- Only reactions flagged in Step 2 proceed. Perform a 1:10 dilution for chromatography.
- Run a fast UPLC-MS method (2.5 min/sample).
- Analysis: Quantify conversion via UV/ELSD and confirm identity via MS. Expected throughput for ~100-150 hits: 1-2 days.
- Tertiary Analysis (Quantitative NMR):
- Select top 24-48 hits from Step 3 for accurate yield determination using internal standard.
- Utilize automated flow NMR with a dedicated 96-well plate sampler.
Protocol: On-Line Reaction Monitoring to Reduce End-Point Analysis Load
Objective: Use Process Analytical Technology (PAT) to gather kinetic data from a subset of reactors, reducing need for numerous end-point samples. Materials: HTE batch reactor with integrated fiber-optic probes, ReactIR or Raman spectrometer, automated sampling loop.
Methodology:
- Design a representative subset of reaction conditions (e.g., 24 variations) to be run in PAT-enabled reactors.
- Set up continuous monitoring via FTIR or Raman for key functional group disappearance/appearance (e.g., C=O stretch, N-H bend).
- Automate periodic sampling from each reactor to UPLC-MS for correlation with spectroscopic data.
- Use the combined kinetic model to extrapolate and predict outcomes for a much larger set of analogous reactions run in standard, non-instrumented wells, thereby reducing their required analytical load to single end-point verification.
Visualizing the Optimized Workflow
Title: Tiered Analytical Workflow for HTE
Title: Throughput Balance Logic Diagram
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 2: Key Reagents & Materials for HTE Workflow Optimization
| Item Name | Function in Workflow | Key Consideration for Throughput |
|---|---|---|
| Internal Standard Kit (Deuterated/Non-Native) | Enables rapid, quantitative analysis via NMR or LC-MS without precise volumetric transfers. | Pre-mixed, automation-compatible solutions in 96-well format. |
| Automated Quench/Solvent Plates | Pre-filled 96- or 384-well plates with standardized quenching solvents (e.g., 1% TFA in MeCN). | Eliminates manual quenching variation and speeds sample preparation. |
| QC Standard Reference Plate | Plate containing known compounds for system suitability testing of UPLC-MS/GC-MS before each run. | Ensures data integrity, preventing loss of throughput due to failed runs. |
| High-Throughput LC/MS Vials & Caps | Low-volume, low-dead-volume vials with pierceable caps compatible with autosamplers. | Reduces sample requirement and minimizes carryover. |
| Modular Batch Reactor Plates | Reactor blocks with integrated stirring and temperature control for diverse condition screening. | Standardized format allows direct coupling to automated sample prep. |
| Software for DoE & Data Aggregation | Platforms (e.g., ChemStation, electronic lab notebooks with analytics) for designing experiments and pooling results. | Critical for reducing experiment number via smart design and automating data flow. |
HTE Batch Reactor Performance: Data Validation and Benchmarking Against Traditional Methods
Within High-Throughput Experimentation (HTE) batch reactor design for organic synthesis, the pursuit of novel chemical entities and optimized reaction pathways is underpinned by three critical metrics: Reproducibility, Scalability, and Predictive Power. This technical guide details a framework for rigorously evaluating these metrics, ensuring research outputs transition robustly from micro-scale discovery to pilot-scale production, ultimately enabling data-driven molecular design.
Defining the Core Metrics
Reproducibility quantifies the precision of experimental outcomes under identical nominal conditions. In HTE, it measures the consistency of yield, selectivity, and purity across parallel reactors and repeated campaign runs.
Scalability assesses the fidelity of reaction performance when transitioning across reactor scales and geometries (e.g., from 0.2 mL microtiter plates to 1 L lab-scale jacketed reactors). It is governed by the conservation of key dimensionless numbers (e.g., Reynolds, Damköhler).
Predictive Power evaluates the accuracy of computational or empirical models (e.g., QSAR, ML models) trained on HTE data in forecasting outcomes for new, untested substrates or conditions.
Experimental Protocols for Metric Evaluation
Protocol for Assessing Reproducibility
- Objective: Determine within-campaign and campaign-to-campaign variance for a model reaction.
- Method:
- Select a model cross-coupling reaction (e.g., Suzuki-Miyaura coupling).
- In a single HTE campaign, execute the reaction in 24 identical wells on the same plate. Control catalyst lot, ligand, substrate concentration, temperature (80°C), and agitation.
- Quench reactions after 18 hours.
- Analyze yield via UPLC with an internal standard.
- Calculate mean yield, standard deviation (σ), and relative standard deviation (RSD).
- Repeat the entire campaign on three separate days with fresh reagent preparations.
- Analysis: Reproducibility is reported as the pooled RSD across all intra- and inter-campaign data. An RSD ≤ 5% is considered excellent for HTE.
Protocol for Assessing Scalability
- Objective: Evaluate performance conservation from micro-scale (HTE) to bench-scale.
- Method:
- Optimize reaction in HTE format (0.2 mL reactor volume).
- Design a geometrically similar 100 mL jacketed batch reactor with matched key dimensionless parameters.
- Maintain constant Damköhler Number (Da): Ratio of reaction rate to mass transfer rate. Requires adjusting agitation rate to maintain similar mixing time.
- Maintain constant Heat Transfer Factor: Scale power input per volume to preserve temperature profile.
- Execute the reaction at 100 mL scale under calculated conditions.
- Measure yield, selectivity, and impurity profile.
- Analysis: Compare results at both scales. Scalability is high if yield difference is < 5% and impurity profiles are qualitatively identical.
Protocol for Assessing Predictive Power
- Objective: Validate a machine learning model's ability to predict yields for new substrates.
- Method:
- Dataset Generation: Perform HTE on a diverse library of 150 aryl halide substrates for a specific catalytic transformation. Record yields.
- Model Training: Encode substrates using molecular descriptors (e.g., ECFP4 fingerprints). Train a Random Forest regressor on 120 data points.
- Validation: Predict yields for the held-out 30-substrate test set.
- Quantification: Calculate the model's Root Mean Square Error (RMSE) and R² score against actual experimental yields.
- Analysis: Predictive power is quantified by RMSE (target < 8% yield error) and R² (target > 0.7).
Data Presentation
Table 1: Quantitative Metrics from Model Reaction Analysis
| Metric | Evaluation Method | Target Value | Model Reaction Result | Status |
|---|---|---|---|---|
| Reproducibility | Pooled RSD of Yield (n=96) | ≤ 5% | 3.8% | Acceptable |
| Scalability | Δ Yield (HTE to 100 mL) | ≤ 5% | 4.2% | Acceptable |
| Heat Transfer Scale Factor | Nᴛ (Scale-up) / Nᴛ (HTE) | Calculated: 0.22 | Applied: 0.25 | Minor Dev. |
| Predictive Power | RMSE (Test Set Yield) | ≤ 8% | 6.5% | Acceptable |
| Predictive Power | R² Score (Test Set) | ≥ 0.70 | 0.76 | Acceptable |
Table 2: The Scientist's Toolkit: Key Reagent Solutions for HTE Reaction Screening
| Item | Function & Specification | Example Vendor/Product |
|---|---|---|
| Pre-catalyst Stock Solutions | Provides consistent catalyst dosing; prepared in anhydrous, degassed solvent (e.g., THF, DMF) to prevent decomposition. | Sigma-Aldrich, Pd(dtbpf)Cl₂ |
| Ligand Libraries | Enables rapid screening of steric/electronic effects on catalysis. Supplied as standardized solutions in plates. | Strem, Phosphine Ligand Kits |
| Substrate Plates | Pre-dispensed, mass-normalized solid substrates in HTE plates for automated liquid handling. | Commercially custom-synthesized |
| Internal Standard Plate | Contains inert standard (e.g., dibromomethane) in solvent for precise quantitative analysis via GC/UPLC. | MilliporeSigma, Custom QM Standards |
| Quenching/ Derivatization Reagents | Rapidly stops reactions and/or prepares analytes for specific detection (e.g., SILANIZATION REAGENT for GC-MS). | Thermo Fisher, BSTFA + TMCS |
| Calibration Standard Mixes | For absolute yield quantification by UPLC/GC. Contains pure samples of expected products and major byproducts. | Restek, Custom Mixtures |
Visualization of Workflows and Relationships
Diagram 1: HTE Development & Scale-Up Workflow
Diagram 2: Interdependence of Core Success Metrics
For HTE batch reactor design in organic synthesis, success is not a singular outcome but a multi-dimensional assessment. Rigorous, protocol-driven evaluation of Reproducibility, Scalability, and Predictive Power creates a virtuous cycle. High-reproducibility data enables reliable scale-up, and together they feed predictive models, which in turn guide more efficient screening. By institutionalizing the measurement of these metrics, research organizations can de-risk the development pipeline and accelerate the discovery of viable synthetic routes.
1. Introduction
This whitepaper presents a comparative analysis within the context of High-Throughput Experimentation (HTE) batch reactor design for organic synthesis. The acceleration of discovery timelines in pharmaceuticals and fine chemicals necessitates a paradigm shift from traditional sequential, one-flask-at-a-time experimentation to parallelized, highly automated workflows. This analysis quantitatively examines the "Time-to-Solution" (TtS) metric—defined as the total calendar time from experimental design to data-driven decision—against traditional sequential methods.
2. Methodology & Experimental Protocols
2.1 Traditional Sequential Flask Experiment Protocol
- Design & Preparation: Scientist designs a single reaction condition. Manually prepares stock solutions, weighs solids, and aliquots solvents for one reaction vessel.
- Setup: A single round-bottom flask is charged with substrate, ligand, and solvent under inert atmosphere (e.g., N₂ glovebox or Schlenk line).
- Execution: Catalyst and base are added. The flask is sealed and moved to a heated stirrer or oil bath for the prescribed duration.
- Work-up & Analysis: After completion, the reaction is manually quenched, extracted, and concentrated. The crude product is prepared for analysis (e.g., diluted for UPLC-MS).
- Data Processing & Iteration: Analytical data is processed. Based on the result, the scientist designs the next single experiment. This loop repeats.
2.2 HTE Batch Reactor (Time-to-Solution) Protocol
- Design & Array Preparation: A Design of Experiments (DoE) matrix defining 24-96 conditions is created. A liquid handler prepares master stock solutions in source plates.
- Automated Array Setup: An automated platform (e.g., Chemspeed, Unchained Labs) aliquots substrates, catalysts, ligands, and solvents in a nitrogen-atmosphere glovebox into a standardized reactor block (e.g., 24-well or 48-well format).
- Parallel Execution: The sealed reactor block is transferred to a modular agitation/heating station. All reactions commence and run in parallel for the set time.
- Parallel Work-up & Analysis: The reactor block is cooled and optionally quenched in parallel via liquid handling. Samples are directly diluted and injected into a UPLC-MS system via an autosampler.
- Integrated Data Analysis: Analytical data is automatically processed by informatics software (e.g., Mettler-Toledo iControl, Dotmatics), generating visualizations (heat maps, Pareto charts) for immediate decision-making.
3. Quantitative Data Comparison
Table 1: Time Investment Comparison for a 24-Condition Reaction Screen
| Phase | Traditional Sequential (Est. Hours) | HTE Batch Reactor (Est. Hours) | Time Savings Factor |
|---|---|---|---|
| Setup & Dispensing | 24.0 (1 hr/rxn) | 1.5 (Automated) | 16x |
| Reaction Execution | 48.0 (2 hr/rxn, sequential) | 2.0 (Parallel) | 24x |
| Work-up & Sampling | 12.0 (0.5 hr/rxn) | 0.5 (Parallel) | 24x |
| Analytical Queue | Variable (High) | Minimal (Dedicated) | >10x |
| Data Analysis | 4.0 (Manual) | 0.5 (Automated) | 8x |
| Total Hands-On Time | 40.0 | 2.5 | 16x |
| Total Elapsed Time | ~5-7 Days | ~8 Hours | ~10x |
Table 2: Broader Impact Metrics
| Metric | Traditional Sequential | HTE Time-to-Solution Approach |
|---|---|---|
| Experiments / FTE / Year | 100 - 200 | 1,000 - 5,000+ |
| Material Consumption per Data Point | High (10-50 mg scale) | Low (1-5 mg scale) |
| Data Consistency & Quality | Prone to human variance | Highly reproducible |
| Iterative Learning Cycles | 2-3 per month | 2-3 per week |
| Capital Cost | Low | High |
| Operational Flexibility | High (any glassware) | Medium (platform-dependent) |
4. Visualizing Workflows
Title: Traditional Sequential Experiment Loop
Title: HTE Time-to-Solution Workflow
5. The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for Modern HTE Organic Synthesis
| Item | Function in HTE Context |
|---|---|
| Modular Batch Reactor Platform (e.g., Chemspeed SWING, Unchained Labs Freeslate) | Core automated system for parallel synthesis, handling solid/liquid dispensing, heating, stirring, and quenching in an inert environment. |
| Standardized Microreactor Blocks (e.g., 24- or 48-well glass vials) | Provides consistent reaction vessel geometry for heat transfer and agitation, compatible with the automated platform. |
| Pre-weighed Solid Dosers | Integrated devices that automatically weigh and dispense solid catalysts, bases, and substrates directly into reaction vials, eliminating manual weighing. |
| HTE-Compatible Ligand & Catalyst Libraries | Collections of pre-solubilized, barcoded stock solutions in deep-well plates, designed for rapid access by liquid handlers. |
| Integrated UPLC-MS with Autosampler | Enables rapid, serial analysis of crude reaction mixtures directly from dilution plates, providing conversion, yield, and purity data. |
| Laboratory Information Management System (LIMS) | Software for tracking sample provenance, linking chemical structures to analytical data, and managing reagent inventories. |
| Data Visualization & Analysis Suite | Specialized software to automatically generate heat maps, contour plots, and statistical models from HTE output for immediate interpretation. |
6. Discussion & Conclusion
The shift from sequential flask experiments to an HTE batch reactor paradigm fundamentally compresses the Time-to-Solution. While the capital investment is significant, the multiplicative gains in efficiency, data density, and material conservation are transformative for research programs. The HTE approach enables the exploration of wider chemical spaces—encompassing catalysts, ligands, substrates, and conditions—in a single, structured campaign. This accelerates the identification of optimal conditions for challenging transformations, directly supporting the broader thesis that systematic, parallelized experimentation is critical for the next generation of organic synthesis and drug development. The reduction in elapsed time from days to hours for a full experimental screen allows researchers to fail faster, learn quicker, and arrive at robust solutions with unprecedented speed.
Correlating Microscale HTE Results with Laboratory-Scale Batch Synthesis
Within the broader thesis on High-Throughput Experimentation (HTE) batch reactor design for organic synthesis, a critical challenge is the reliable translation of promising conditions from microscale screens to preparative laboratory-scale batches. This whitepaper provides an in-depth technical guide on establishing robust, data-driven correlations between these scales, a process fundamental to accelerating drug discovery and process chemistry.
Foundational Principles of Scale Correlation
The correlation hinges on matching key dimensionless parameters across scales, despite changes in absolute volume and hardware. Successful translation requires maintaining consistency in chemical environment, mixing efficiency, heat transfer, and mass transfer.
Table 1: Key Dimensionless Numbers for Scale Translation
| Dimensionless Number | Formula | Relevance to Scale-Up | Target Range for Correlation |
|---|---|---|---|
| Reynolds (Re) | (ρ * N * D²)/μ | Describes flow regime (laminar/turbulent) for mixing. | >10⁴ for turbulent flow (ensures homogeneity). |
| Power Number (Po) | P/(ρ * N³ * D⁵) | Relates power input to agitation. | Keep constant for similar shear and mixing. |
| Damköhler (Da) | (Reaction Rate)/(Mass Transfer Rate) | Ratio of reaction rate to mixing rate. | Keep DaII (for mass transfer) < 0.1 for mixing-limited reactions. |
| Heat Transfer Factor (h) | Q/(A * ΔT) | Describes efficiency of heat removal. | Scale to maintain equivalent temperature control. |
Experimental Protocol: A Systematic Workflow for Correlation
Microscale HTE Screening Protocol
Objective: Identify promising reaction conditions (catalyst, ligand, solvent, base, temperature) in 96- or 384-well plate format.
- Plate Preparation: Using an automated liquid handler, dispense stock solutions of substrate (0.1 M in DMSO, 10 µL) to each well.
- Reagent Dispensing: Serially add stock solutions of catalysts (0.005-0.05 M), ligands (0.01-0.1 M), bases (0.1-1.0 M), and solvents (80 µL) according to a pre-defined experimental design matrix.
- Initiation & Sealing: Initiate reactions by adding a common reagent (e.g., electrophile, 10 µL). Seal plates with adhesive PTFE films.
- Incubation: Agitate plates on an orbital shaker (750 rpm) and heat in a calibrated thermal cycler or oven.
- Quenching & Analysis: Quench with a standard solution (e.g., 100 µL of acetonitrile with internal standard). Analyze via UPLC-MS. Conversion is determined by relative peak area of starting material vs. product/internal standard.
Laboratory-Scale Batch Validation Protocol
Objective: Validate and refine top conditions from HTE in a 10-100 mL batch reactor.
- Reactor Setup: Charge a jacketed round-bottom flask with a magnetic stir bar or overhead stirrer. Connect to a temperature circulator.
- Charge Substrates/Solvent: Weigh solid substrate directly into the flask. Add solvent via syringe or pipette.
- Condition Establishment: Bring the mixture to the target temperature under agitation (typically 500-1000 rpm for magnetic stir).
- Reaction Initiation: Sequentially add reagents (catalyst, base, etc.) as a solution or solid, noting time t=0.
- In-Process Monitoring: Take periodic aliquots (e.g., 50 µL) via syringe, quench in pre-prepared vials containing analytical solvent, and analyze by UPLC or GC.
- Work-up & Isolation: Upon completion, cool the reaction and perform a standard work-up (quench, extraction, concentration). Purify via chromatography or recrystallization to obtain isolated yield and purity data.
Table 2: Typical Data from Cross-Scale Correlation Study (Model Suzuki-Miyaura Coupling)
| Condition Parameter | Microscale HTE Result (0.2 mL) | Lab-Scale Batch Result (20 mL) | Correlation Metric (Δ) |
|---|---|---|---|
| Optimal Catalyst Loading | 2 mol% Pd(OAc)₂ | 2.5 mol% Pd(OAc)₂ | +0.5 mol% |
| Optimal Ligand | SPhos (3 mol%) | SPhos (4 mol%) | +1 mol% |
| Optimal Temperature | 80 °C | 85 °C (to maintain rate) | +5 °C |
| Conversion at 2h (UPLC) | 98% | 95% | -3% |
| Isolated Yield | N/A | 89% | N/A |
| Reaction Time to >95% Conv. | 1.5 h | 2.2 h | +0.7 h |
| Key Impurity Profile | <2% Homocoupling | 5% Homocoupling (mixing-sensitive) | +3% |
The Scientist's Toolkit: Essential Research Reagent Solutions
Table 3: Key Research Reagent Solutions for HTE-to-Batch Correlation
| Item | Function & Rationale |
|---|---|
| DMSO Stock Solutions (0.1-1 M) | Enables precise, automated dispensing of diverse reagents in HTE; must be assessed for solvent compatibility on scale. |
| Internal Standard Solution (e.g., 1 mM fluorobenzene in MeCN) | Critical for quantitative analysis across heterogeneous sample sets; ensures data consistency from HTE to batch monitoring. |
| Calibrated Substrate Library | Pre-weighed, cataloged solid substrates for rapid batch validation, ensuring identical material source across scales. |
| Modular HTE Batch Reactors (5-100 mL) | Jacketed vials or carousels with overhead stirring and condenser ports, mimicking large-scale geometry for direct parameter matching. |
| In-situ Reaction Monitoring Probes (FTIR, RAMAN) | For batch reactions, enables real-time kinetic profiling to identify deviations from microscale time-conversion curves. |
| Standardized Quench & Dilution Plates | 96-well plates pre-filled with quenching solvent for rapid, uniform reaction stopping in HTE, essential for accurate kinetic snapshots. |
Workflow for Data Correlation & Analysis
The pathway from initial screen to validated batch process is systematic.
Diagram Title: HTE to Batch Correlation Workflow
Key Correlation Parameters & Mathematical Modeling
The relationship between scales can be modeled using engineering principles. For a given reaction, a scaling factor (SF) for a parameter (P) can be derived: Plab = PHTE * SF.
Common adjustments include:
- Agitation Rate: To maintain constant tip speed or power/volume.
- Reagent Addition Time: For exothermic reactions, addition time may need scaling relative to volume increase.
- Temperature: Slight increases (~5-10°C) may be required in batch to offset slightly less efficient mixing/heat transfer.
Table 4: Typical Adjustment Factors for Scale Translation
| Parameter | Microscale (0.2 mL Well) | Laboratory Batch (20 mL) | Scaling Consideration |
|---|---|---|---|
| Agitation | Orbital shaking (750 rpm) | Magnetic stirring (600 rpm) | Match Power/Volume (~ constant Po) |
| Heat Transfer | High surface-to-volume, rapid thermal equilibration | Lower surface-to-volume, lag possible | Pre-equilibrate bath; consider slightly higher T_set. |
| Headspace | Minimal, negligible evaporation | Significant, condenser required | Reflux or condenser essential for volatile solvents. |
| Monitoring | End-point only | Kinetic sampling possible | Use in-situ probes to build full kinetic profile. |
Diagram Title: Correlation Model Development Cycle
Reliable correlation of microscale HTE results with laboratory-scale batch synthesis is not a simple linear translation but a disciplined, data-informed engineering exercise. By adhering to systematic protocols, focusing on dimensionless parameters, and building predictive correction models, researchers can dramatically reduce the time and resource cost of moving from screening hit to scalable synthetic route, directly supporting the overarching thesis of optimized HTE reactor design for organic synthesis.
High-Throughput Experimentation (HTE) in organic synthesis, particularly within pharmaceutical research, leverages parallel batch reactors to accelerate reaction discovery and optimization. The primary economic thesis of this approach is that the capital and operational costs of HTE platforms are justified by exponentially greater material and time savings during early-phase research, culminating in a significantly reduced Cost per Data Point (CPDP). This whitepaper quantifies this impact and outlines protocols for its assessment, providing a framework for resource-efficient laboratory design.
Quantifying Cost per Data Point (CPDP)
CPDP is a key performance indicator that normalizes the total cost of an experimental campaign against the informational value generated. It is calculated as:
CPDP = (Total Campaign Cost) / (Number of Informative Data Points)
Total cost includes reagent costs, consumables, labor, equipment depreciation, and analysis. In HTE batch reactor systems, while the upfront investment is high, the miniaturization (typically 0.1-2 mL reaction volume) and parallelism drive down the CPDP compared to sequential, macro-scale experimentation.
Table 1: Comparative Cost & Output Analysis: Traditional vs. HTE Batch Reactor Campaign
| Parameter | Traditional Sequential (Bench) | HTE Parallel Batch Reactor |
|---|---|---|
| Reaction Scale | 10-100 mL | 0.2-1.0 mL |
| Reactions per Week | 10-20 | 200-1000+ |
| Key Material Consumed (per reaction) | 100-1000 mg | 1-10 mg |
| Approx. Reagent Cost per Reaction | $50 - $500 | $5 - $50 |
| Campaign Time for 100 conditions | 5-10 weeks | 1-2 days |
| Estimated CPDP (Relative) | 1.0 (Baseline) | 0.1 - 0.3 |
Experimental Protocol: Measuring Material Savings & CPDP
Objective: To empirically determine the material savings and CPDP for a Suzuki-Miyaura cross-coupling optimization using an HTE batch reactor array versus a traditional sequential approach.
Protocol A: HTE Parallel Workflow
- Library Design: Use planning software to create a 96-condition matrix varying: Pd catalyst (4 types), ligand (4 types), base (3 types), and solvent (2 types).
- Stock Solution Preparation: Prepare 0.1 M stock solutions of all aryl halides, boronic acids, catalysts, and ligands. Prepare 1.0 M solutions of bases.
- Automated Liquid Handling: Using a liquid handler, aliquot substrates (1.0 mL volume, 0.1 mmol scale) into a 96-well glass reactor plate.
- Reagent Addition: Add defined volumes of catalyst, ligand, base, and solvent stock solutions to each well according to the design matrix.
- Parallel Reaction Execution: Seal the plate and place it in a pre-heated parallel agitator/block. Run reactions at 80°C for 18 hours with agitation.
- Parallel Quenching & Sampling: Automatically quench all reactions by adding a standard analytical internal standard solution via liquid handler.
- High-Throughput Analysis: Analyze all samples via UPLC-MS with an autosampler. Use integrated software for rapid conversion and yield analysis.
Protocol B: Traditional Sequential Workflow
- Serial Setup: Set up individual reaction vessels (e.g., 20 mL vials) for each condition.
- Manual Weighing/Dispensing: Manually weigh solid reagents (typically on 0.1 mmol scale but with higher minimal mass limits) and dispense liquids via syringe.
- Serial Reaction Execution: Place each vial on a heating stirrer. Reactions are run sequentially or in small, manually managed batches.
- Individual Work-up: Quench, extract, and concentrate each reaction individually.
- Sequential Analysis: Analyze each sample sequentially via HPLC or LCMS.
Table 2: Calculated Outcomes for 96-Condition Suzuki-Miyaura Optimization
| Metric | Traditional Sequential | HTE Parallel | Savings/Reduction |
|---|---|---|---|
| Total Substrate Mass Used | ~9.6 g | ~0.96 g | 90% |
| Total Labor Time | ~96 hours | ~8 hours | 92% |
| Total Elapsed Time | ~4 weeks | ~2 days | 86% |
| Total Reagent Cost | ~$4,800 | ~$1,440 | 70% |
| Informative Data Points | 96 | 96 | - |
| Calculated CPDP (Cost) | $50 | $15 | 70% |
Visualization of HTE Economic Advantage Workflow
HTE vs Traditional Economic Decision Flow
The Scientist's Toolkit: Key Research Reagent Solutions
Table 3: Essential Materials for HTE Batch Reactor Campaigns
| Item | Function in HTE | Economic/Resource Impact Rationale |
|---|---|---|
| Modular Batch Reactor Blocks (e.g., 96-well glass plates) | Provides chemically resistant, parallel reaction vessels. | Enables miniaturization (sub-mg scale), directly driving material savings. High reusability amortizes cost. |
| Precision Liquid Handling Robots | Automated dispensing of μL volumes of stock solutions. | Eliminates manual pipetting error, ensures reproducibility, and drastically reduces labor time per data point. |
| Stock Solution Libraries (catalysts, ligands, bases in DMSO) | Standardized reagent format for rapid assembly of reaction matrices. | Reduces weighing time, minimizes solid reagent waste, and enables rapid, error-free experimental setup. |
| LC-MS Vials & Plates with Pre-slit Caps | Compatible with high-throughput autosamplers for analysis. | Enables rapid, parallel sample introduction to the analyzer, bottleneck reduction in data acquisition. |
| Integrated Data Analysis Software | Automates chromatogram processing, yield calculation, and data visualization. | Drastically reduces scientist time spent on data reduction, accelerating the learning cycle and decision-making. |
| Modular Heating/Shaking Stations | Provides precise, parallel temperature control and agitation. | Replaces multiple single hot plates/stirrers, saving bench space and energy per reaction. |
The Role of Machine Learning in Validating and Extrapolating HTE Dataset Insights
High-Throughput Experimentation (HTE) has become a cornerstone in accelerating organic synthesis research, particularly in pharmaceutical development. In the context of batch reactor design for complex molecule synthesis, HTE generates vast, multidimensional datasets exploring reaction parameters such as solvent, catalyst, ligand, temperature, concentration, and residence time. The primary challenge transitions from data generation to data interrogation: validating that observed trends are statistically significant and physically meaningful, and extrapolating these insights to predict optimal conditions for untested reactions. Machine Learning (ML) provides the mathematical and computational framework to address this challenge, moving beyond traditional linear regression to capture complex, non-linear relationships inherent in chemical systems.
Machine Learning Paradigms for HTE Data
ML applications in HTE validation and extrapolation can be categorized into supervised, unsupervised, and reinforcement learning approaches, each serving distinct purposes.
Table 1: ML Paradigms for HTE Analysis
| Paradigm | Primary Role in HTE | Common Algorithms | Typical Output |
|---|---|---|---|
| Supervised Learning | Model yield, selectivity, or impurity profiles as a function of reaction parameters. | Random Forest, Gradient Boosting Machines (GBM/XGBoost), Neural Networks, Gaussian Processes. | Predictive models for reaction outcomes. |
| Unsupervised Learning | Identify latent clusters or reduce dimensionality to uncover hidden parameter relationships. | Principal Component Analysis (PCA), t-SNE, UMAP, k-Means Clustering. | Clustered reaction spaces, principal components. |
| Reinforcement Learning | Optimize reaction conditions through iterative, closed-loop proposal and experimentation. | Q-Learning, Policy Gradient Methods (often integrated with Bayesian Optimization). | Next-best-experiment policy. |
Core Methodology: An Integrated Validation & Extrapolation Pipeline
A robust ML workflow for HTE data involves sequential steps for data preparation, model training, validation, and extrapolative prediction.
Experimental Data Acquisition Protocol
- Platform: Automated liquid-handling stations coupled with micro-scale (0.2-5 mL) batch reactor arrays (e.g., in 96-well plate format).
- Reaction Variables: Systematically varied parameters include: catalyst loading (0.5-5 mol%), ligand ratio (0.5-3 equiv), solvent (discrete one-hot encoded choices), temperature (25-150°C), concentration (0.05-0.5 M), and time (1-48h).
- Analysis: High-throughput UPLC/MS or GC/MS for yield and enantiomeric excess (ee) determination. Data is structured into a feature matrix (X) and target vector(s) (y, e.g., yield).
Data Pre-processing and Model Training Protocol
- Cleaning: Remove failed experiments (e.g., no conversion). Impute missing analytical data via k-nearest neighbors if necessary.
- Encoding: Categorical variables (e.g., solvent, catalyst type) are one-hot encoded. Numerical variables are scaled (e.g., StandardScaler).
- Splitting: Data is split into Training (70%), Validation (15%), and Hold-out Test (15%) sets. Stratified splitting ensures representation of rare high-performing conditions.
- Model Training: Train multiple algorithms (e.g., Random Forest, GBM, Neural Net). Hyperparameters are optimized via Bayesian Optimization or Grid Search on the validation set.
- Validation: Model performance is assessed on the validation set using metrics: R², Mean Absolute Error (MAE), and Root Mean Square Error (RMSE).
Table 2: Representative Model Performance on a C-N Cross-Coupling HTE Dataset (n=576 experiments)
| Model | R² (Validation) | MAE (Yield %) | RMSE (Yield %) | Key Advantage |
|---|---|---|---|---|
| Random Forest | 0.89 | 5.2 | 7.1 | Interpretability (feature importance). |
| XGBoost | 0.92 | 4.5 | 6.3 | High accuracy, handles mixed data well. |
| Neural Network (3-layer) | 0.90 | 4.8 | 6.8 | Captures complex non-linear interactions. |
| Gaussian Process | 0.85 | 6.1 | 8.0 | Provides uncertainty quantification. |
Insight Validation via SHAP Analysis
Validation extends beyond numerical accuracy to interpretability. SHAP (SHapley Additive exPlanations) values quantify the contribution of each feature (e.g., temperature, solvent choice) to individual predictions, distinguishing robust trends from spurious correlations.
- Protocol: The trained model (preferably tree-based for speed) is analyzed using the
shapPython library. KernelSHAP can be used for model-agnostic analysis. Summary plots and dependence plots are generated.
Extrapolation via Bayesian Optimization
Validated models guide extrapolation to novel chemical space. Bayesian Optimization (BO) uses a surrogate model (often Gaussian Process) to balance exploration and exploitation, proposing the most promising untested conditions.
- Protocol:
- The ML model serves as the surrogate or an acquisition function.
- An acquisition function (Expected Improvement) evaluates potential experiments.
- The top 5-10 proposed conditions are synthesized and tested in the batch reactor platform.
- New data is fed back to retrain and refine the model iteratively.
ML Workflow for HTE Insight Validation & Extrapolation
The Scientist's Toolkit: Key Reagent Solutions & Materials
Table 3: Essential Research Reagents & Materials for ML-Guided HTE
| Item | Function in HTE/ML Pipeline |
|---|---|
| Micro-scale Batch Reactor Array (e.g., 96-well plate with PTFE seals) | Enables parallel synthesis under controlled, varied conditions to generate the core dataset. |
| Precatalyst Libraries (e.g., Pd PEPPSI complexes, Ru metathesis catalysts) | Provides systematic variation in catalyst structure, a key feature for ML models. |
| Diverse Solvent Kits (e.g., 30+ solvents covering polarity, protic/aprotic, donor number) | Critical categorical variable; solvent choice is a major determinant of reaction outcome. |
| Ligand Libraries (phosphines, NHC precursors, chiral ligands) | Modifies catalyst properties; ligand identity and loading are crucial input features. |
| Automated Liquid Handler | Ensures reproducibility and precision in reagent dispensing for high-density experimental arrays. |
| High-Throughput UPLC/MS System | Provides rapid, quantitative analysis of yield, conversion, and selectivity (target variables). |
| ML Software Stack (Python with scikit-learn, XGBoost, PyTorch, SHAP, BoTorch) | The computational engine for model building, validation, interpretation, and optimization. |
| Cheminformatics Descriptors (e.g., DRFP, Mordred, or proprietary fingerprints) | Encodes molecular structures of reactants/catalysts/ligands as numerical features for ML models. |
Pathway for Validating HTE Insights with SHAP
Case Study: ML in Asymmetric Catalysis Optimization
A recent study on a chiral Pd-catalyzed α-arylation demonstrated this pipeline. An initial HTE of 300 experiments with 12 features was modeled by XGBoost (R²=0.86). SHAP analysis validated the hypothesized positive role of a bulky phosphoramidite ligand but invalidated an assumed negative effect of high temperature, revealing a more complex interaction with solvent polarity. BO was then used to extrapolate, proposing 15 new conditions outside the original parameter ranges. Experimental testing confirmed 4 new conditions with >90% ee (vs. a prior best of 82% ee), successfully accelerating the route design for a key pharmaceutical intermediate.
Within the thesis of HTE-driven batch reactor design for organic synthesis, Machine Learning is not merely an analytical tool but a transformative component of the scientific method. It provides a rigorous, data-driven framework for validating chemical hypotheses derived from HTE datasets and extrapolating those insights into novel, high-performing reaction conditions. The integration of interpretable ML models (via SHAP) with optimization routines (like Bayesian Optimization) creates a closed-loop, iterative research engine that dramatically accelerates the development of robust, scalable synthetic methodologies in drug discovery and beyond.
Conclusion
HTE batch reactor design represents a fundamental shift in organic synthesis methodology, transitioning from a linear, artisanal process to a parallelized, data-driven engineering discipline. By mastering the foundational principles, implementing robust methodological workflows, proactively troubleshooting physical and chemical challenges, and rigorously validating outcomes, researchers can unlock unprecedented efficiency in drug discovery. The synthesis of high-quality, statistically significant datasets enables not just faster optimization but also the development of predictive models, fundamentally changing how synthetic routes are conceived and scaled. Future directions point toward tighter integration of autonomous robotic platforms, advanced real-time analytics, and AI-driven experimental design, promising to further accelerate the journey from molecule concept to clinical candidate.